Figure 1
Lecturer: Kenneth Korr, MD
GENERAL PRINCIPLES OF VALVE MALFUNCTION:
VALVULAR STENOSIS:
Hemodynamic Hallmark = "PRESSURE GRADIENT"
Hemodynamic Hallmark = "VOLUME OVERLOAD"
EXAMPLES OF VALVULAR HEART DISEASE
MITRAL STENOSIS:
Pathophysiology:
Obstruction of blood flow from LA to
LV during diastole, causing increased pressure in the left atrium, pulmonary
capillaries and, eventually, the right side of the heart. (see fig
1 and fig 1A)
As the valve area (i.e. the cross-sectional
area of the valve opening during diastole) becomes smaller, the pressure
gradient increases. The relationship between the mitral valve area, the
forward cardiac output and the pressure gradient across the valve during
diastole is complex, and is defined by this equation:
| CARDIAC OUTPUT (mI/min) | ||
| MITRAL VALVE AREA | = | DIASTOLIC FILLING PERIOD
(sec/min)
|
| SQ ROOT of PRESSURE GRADIENT (mm Hg) |
Examine the relationship between mitral valve gradient and flow (cardiac output) for various valve areas. (Fig 2) As valve area gets smaller, conditions which increase valve flow (exercise, tachycardia) result in an increase in LA pressure (and hence worsening symptoms i.e. DYSPNEA).
Symptoms:
Dyspnea, Orthopnea, PNDPhysical Signs:
Cough & Hemoptysis
Atrial Fibrillation, LA thrombus and systemic Embolization
RV failure, TR and systemic congestion
Diastolic murmur (low-pitched rumble) & Opening Snap
Treatment:
Medical Rx:
Anticoagulants -- prevent systemic embolization
Diuretics -- reduce pulmonary vascular
congestion Surgical Rx
MITRAL REGURGITATION:
1) the size of the mitral orifice during regurgitation
2) the systemic vascular resistance opposing forward flow from the ventricle
3) the compliance of the left atrium
4) the systolic pressure gradient between the LV and the LA
5) the duration of regurgitation during systole (not all regurgitation is holo-systolic)
Effect of MR on LA pressure
depends on LA Compliance (see Figure 3)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Anatomic structures integral to MV competence:
Myxomatous degeneration (MV prolapse)
Infectious endocarditis (acute and chronic)
Chronic rheumatic heart disease
Marked LV enlargement from any cause (e.g. dilated cardiomyopathy)
Ruptured papillary muscle &/or chordae tendinae
Hypertrophic Cardiomyopathy with obstruction
Mitral annular Ca++
Congenital cleft MV, etc.
Acute MR: Pulmonary Congestion
Physical findings:
Holosystolic apical murmurTreatment:
+ S3 gallop
+ laterally displaced apical impulse
Medical Rx:
Anticoagulation to prevent systemic embolization
Diuretics to reduce pulmonary congestion
Vasodilators to reduce afterload (impedance to LV ejection)
Surgical Rx: Mitral
valve replacement or repair
AORTIC STENOSIS:
Pathophysiology:
Obstruction to LV outflow during systole.
Pressure gradient across the aortic
valve (pressure higher in LV than aorta during systole), causes chronic
LV "Pressure Overload".
Compensatory concentric left ventricular
hypertrophy --> reduced LV compliance.
Thus, LV is "stiff" (noncompliant)
and LVEDP rises rapidly with increases in LV end-diastolic volume.
|
|
||
| AORTIC VALVE AREA | = | SYSTOLIC EJECTION PERIOD sec/min
|
| SQ ROOT OF SYSTOLIC PRESSURE GRADIENT |
Systolic Ejection Period << Diastolic Filling Period
Thus Aortic Stenosis has higher pressure
gradients and valve flow than Mitral Stenosis (see Fig
4)
DYSPNEA ON EXERTION and CHF
SYNCOPE
AORTIC INSUFFICIENCY:
Pathophysiology:
Compensatory Mechanisms include:
"Eccentric" LV Hypertrophy (dilation
and hypertrophy)
Acute Al -- Acute LV failure and Pulm Edema (without time for LV dilatation). Often refractory to Med Rx and requires emergency aortic valve replacement.
Diseases of Aortic valve leaflets:
Bicuspid Aortic Valve
Infectious Endocarditis
Diseases of the Aortic Root:
Syphilis
Ankylosing Spondylitis and other Connective Tissue Disorders (Reiters Synd, Rheum Arth, SLE, etc.).
Trauma and Aortic Dissection
Physical signs:LV failure: dyspnea, orthopnea, etc.
Widened pulse pressure (diastolic blood pressure is less than half of the systolic blood pressure: e.g.: BP = 140/50 )
Decrescendo Diastolic Murmur -- Increases with increased SVR (handgrip, squatting)
Austin Flint murmur -- diastolic rumble (differentiate from murmur of mitral stenosis).
Medical Rx: Vasodilators -- Dec systemic vascular resistanceDigitalis and Diuretics - once heart failure ensues
Surgical Rx: Aortic valve replacement (timing of valve replacement crucial to prevent irreversible LV failure.
Figure 1

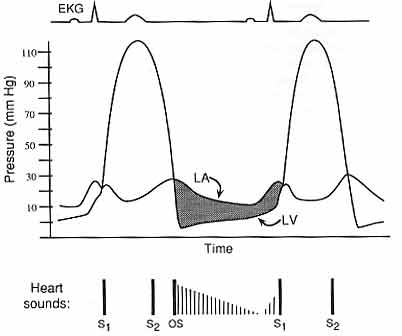
Fig 1: Hemodynamic profile of mitral stenosis. The left atrial (LA) pressure is elevated, and there is a pressure gradient (shaded area) between the LA and left ventricle (LV) during diastole. Abnormal heart sounds are present: there is a diastolic opening snap (OS) that corresponds to opening of the mitral valve, followed by a decrescendo murmur. There is accentuation of the murmur just before S1, due to the increased pressure gradient when the LA contracts. EKG, electrocardiogram.
Figure 1a: Pathophysiology of mitral stenosis (MS)
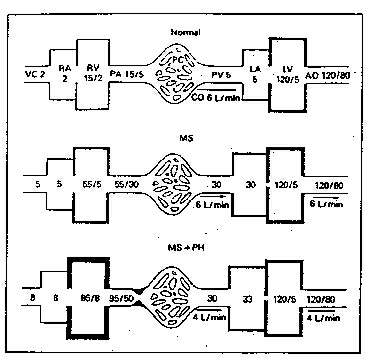
The top panel demonstrates normal hemodynamics. The middle panel shows
a patient with severe mitral stenosis but without reactive pulmonary hypertension
(PH). Left atrial pressure is 30mm Hg and there is a 25-mm gradient (30
- 5 mm Hg) across the mitral valve during diastole. The increased left
atrial pressure is transmitted to the pulmonary capillaries (PC) and pulmonary
artery (PA) and results in an increase in pulmonary arterial pressure to
55/30 mm Hg. Right ventricular systolic pressure must therefore increase
to 55 mm Hg as well. There is slight dilatation and hypertrophy of the
right ventricle (RV) and left atrium (LA). In the lower panel is depicted
an individual with severe reactive pulmonary hypertension. Mitral stenosis
is no more severe than in the patient depicted in the middle panel. However,
a pulmonary hypertensive reaction has developed. The right ventricle is
hypertrophied and dilated. The right ventricle has failed with right atrial
and central venous pressures rising to 8 mm Hg and cardiac output falling
to 4 liters/minute.
VC = vena cava; RA = right atrium; PV = pulmonary veins; LV = left
ventricle; AO = aorta: CO = cardiac output.
Figure 2
Flow Rate (ml/sec)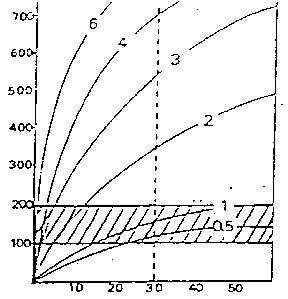
Diastolic Pressure Gradient
(mm Hg)
Relationship between mitral valve gradient, flow rate and different valve areas.
The crosshatched area indicates the range of normal resting flow values. The vertical line represents the threshold for developing pulmonary edema. The pressure gradient increases as flow rate increases, to a small degree with a normal mitral valve (area = 4 - 6 cm2), to a greater degree with a stenotic valve. With severe stenosis, a substantial gradient is present even at rest.
Figure 3 -- Pathophysiology of acute and chronic mitral regurgitation
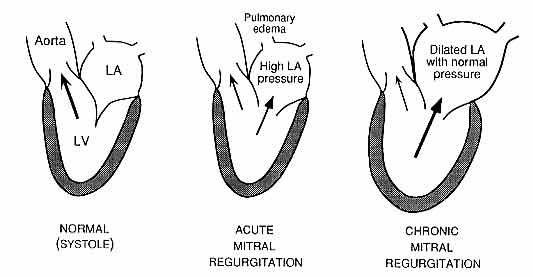
Fig 3: Pathophysiology of mitral regurgitation. In the normal heart, left ventricular (LV) contraction during systole forces blood exclusively through the aortic valve into the aorta; the closed mitral valve prevents regurgitation into the left atrium (LA). In mitral regurgitation (MR), a portion of the LV output is forced retrograde into the LA, so that forward cardiac output into the aorta is reduced. In acute MR, the LA is of normal size and is noncompliant, such that the LA pressure rises markedly and pulmonary edema may result. In chronic MR, the LA has enlarged and is more compliant, such that LA pressure is less elevated and pulmonary congestive symptoms are less common if LV contractile function is intact. There is LV enlargement and eccentric hypertrophy due to the chronic increased volume load.
Figure 4 -- Hemodynamics of aortic stenosis: Relationship between
valve flow rate and mean systolic pressure gradient for various aortic
valve areas. The cross-hatched area indicates the range of normal resting
flow rates. The vertical dashed line indicates a pressure gradient of 50-mm
Hg. Low pressure gradients are noted with normal valve areas even at high
flow rates. Significant reduction in valve area results in substantial
pressure gradients with normal or even depressed values of resting flow.
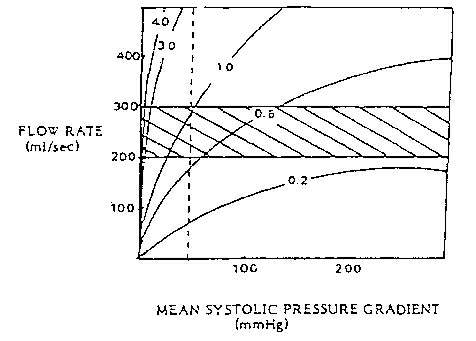
Figure 5.
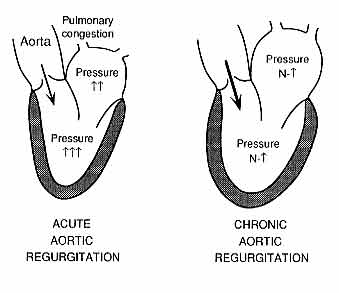
Pathophysiology of acute and chronic aortic regurgitation (AR).
Abnormal regurgitation of blood from the aorta into the left ventricle (LV) is shown in each schematic drawing (large arrows). In acute AR, the LV is of normal size and relatively low compliance, such that its diastolic pressure rises markedly; this is reflected back to the left atrium (LA) and pulmonary vasculature, resulting in pulmonary congestion or edema. In chronic AR, adaptive LV and LA enlargement have occurred, such that a greater volume of regurgitation can be accommodated with less of an increase in diastolic LV pressure, so that pulmonary congestion is less likely. N, normal.