Lab VT
Velocity-Tension relationship in leg muscle demo on a stationary tandem bike
Background: Information in the lecture notes about Hill's equation relating muscle tension to velocity of shortening shows that muscles "doing negatives" (moving in the opposite direction to which they should be contracting) are stronger per fiber than muscles doing positive work. See diagram at bottom of these notes. As noted below, in 1952 A. V. Hill used a modified tandem bicycle to show that a "weak" person pedaling backwards and resisting a "stronger" person will, after a few minutes, exhaust the strong person and be able to reverse the rotation of the gear-linked pedals. Here you and your lab partner will attempt to repeat the demonstration while you monitor tidal volume and concentrations of CO2 and O2 in the exhaled breaths.
"Every Breath You Exhale": Tidal volume refers to the amount of exhaled gas from one breath, measured in liters, and referenced to sea level. Search the web briefly to see various methods of measuring tidal volume. Here you will integrate the output of a flow meter to calculate tidal volume as you breathe into the flow pathway.
WRIT Quiz: What's the difference between "breath" and "breathe"?
1. First let's take up where you might have left off with Lab 9Q: Here we want to regulate the cycles per minute of pedaling to 30.
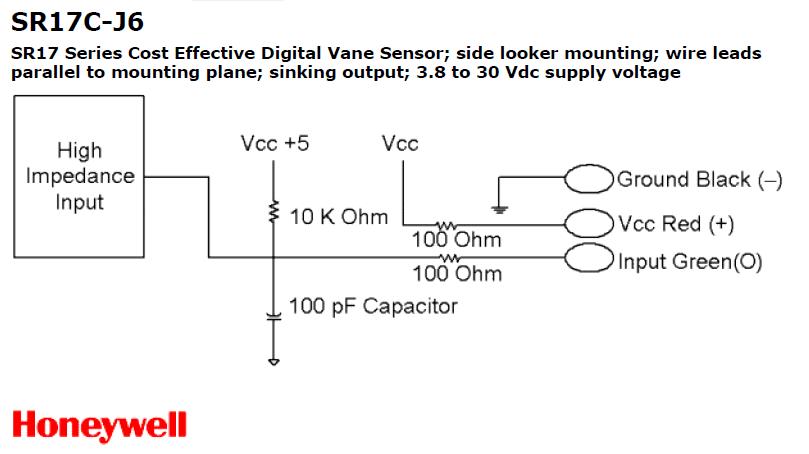
Next to the gear teeth of the idler arm are two Hall-effect
rotation sensors (Honeywell vane sensor SR17C-J6). The sensors are wired as
shown below.
Be careful stepping near the bottom of the tandem bike frame; we don't want any of the wires or resistors down there coming loose out of the breadboard...
You should verify that there are 36 teeth on the pedal sprocket and 12 teeth on the sensing gear.
Going up from the idler gear circuit to the LabView green connector board are 4 wires: Red +5, Black GND and Green and Blue are the two outputs; you should run Green and Blue into the 'scope and see something like the waveforms below, when you turn the wheel. Next, send the 2 digital inputs to the 68-pin LabVIEW connector card, to be read as a scan. By sensing the time between ferromagnetic gear teeth causing Hall effect pulses you can calculate the pedaling rate in rotations per minute. What we want here is 30/min.
Notice that channel A can have a rising edge either when Ch B is Low or Ch B is Hi, depend on CW or CCW rotation. Taking account of this information only will lead to an algorithm that makes mistakes when the wheel jitters back and forth.

As you know from 9Q, it is possible to increase the accuracy and resolution of the two Hall Effect sensors by a factor of 4 over the apparent limit set by angular distance between the gear teeth. Notice that there are 4 edges--A rising, B rising, A falling, B falling--in one "cycle" and each of those edges could increment a counter keeping track of gear angular position. Design a VI that reads on the Hall Effect sensors (perhaps as a scan) and that keeps track of the position of a slowly moving 36-tooth gear. Arrange that turning the gear through one revolution results in a position count of 12*4*3 = 144 or so inside your code. We want output on your front panel in RPM.
Be able to keep track of direction of rotation.
To begin with, have rotation sensing be a separate VI. For a higher sample rate, you may want to scan the two digital channels.
2. Gas Analyzer and Flowmeter:
On and next to the middle table in 097 see the following:
a. Clean green mouthpiece with filter and optional nose clip hanging from the
tandem bike frame
b. Vapor trap through Erlenmeyer flask
www.humboldtmfg.com
c. Omega flow meter model
FMA1845, connected by D-9 sockets to 12v power supply
Users Manual for flowmeter is at http://www.omega.com/manuals/manualpdf/M1680.pdf
where you can see the functions of the 9 pins of colored wires below:
123 D9 socket color code:
5. BLU
9.WHT
4.VIO
8.GRY
3.RED
7.YEL
2.ORJ
6.BRN
1.BLK
d. Removable-for-each-student green mouthpiece, filter and black one-way valve
for release of excess breath.
e. Infrared Industries IR-208 CO2
/ O2 meter, with analog output from back
(C1 is CO2; C2 is O2, on back connector of
IR-208),
and built in filter, pump, and flow control.
 www.infraredindustries.com
www.infraredindustries.com
f. Connections from flowmeter and CO2 monitor to LabVIEW green 68-screw terminal
card:
As set up, on LabVIEW analog-in channels,
ACh 5--CO2
ACh 6--O2
ACh 7--flow meter 0-5v analog
The Omega flow meter LCD display reads full scale 1000L/minute, and the same for its analog 5v output.
1000/60 = 16.6L/sec Full Scale. If reading is 1v, then 16.6/5 = 3.2L/sec
Thus an instantaneous reading of 1V sampled over an interval of 50msec would mean 0.05*3.2 = 0.016 L in that interval. Summing by "for loop shift register" over such intervals would give you approx tidal volume of one exhale in Liters. (Integrate flow). Account for small offset from zero of the flow meter. How can you insure all the breath (or a certain fraction) goes through the meter?
Do not turn off the CO2 meter or the flow meter...they take considerable time to reinitialize...
3. With inlet and outlet of flowmeter disconnected, see what the flowmeter reads... it should be close to 0000. An Agilent 34401 DMM set to DC volts reads the analog out of the flowmeter: what is its zero-flow reading, in mV?
4. With the flowmeter disconnected from the gas analyzer, but with the vapor trap still in place, Create a VI that can calculate the tidal volume of one breath. The VI will start collecting flow data when the flow rate is greater than the zero level seen in part 1. The VI will stop collecting data when the flow rate drops back to the zero level. In your VI you will convert volts out from the flowmeter to L/sec of flow. The VI will record the flow at a reasonable sample rate you determine. The time between samples (sample period) will be used to calculate flow rate for that interval and add that result to a total by way of a LV shift register, if nothing else. Have your VI display flow as a function of time.
5. Arrange that the VI of part 2 can be extended to measure 16 breaths in a row for tidal volume, and display the result on a graph. No nose clip: inhale through your nose. Make sure that after each breath the integrator is "zeroed" and starts over for the next exhale.
4. Take a breath and hold it for 10 seconds. Start your VI running simultaneously with exhaling the breath into your mouthpiece; the VI display should show you the "impulse response" of the CO2 monitor, including the transport delay for coming off 00.00% setting. What is the maximum CO2 from one breath passing through the system? Do a second trial after you set the 208 flowmeter to 2.5 instead of the standard 5.0. How much more transport delay is there? What is the flow rate through the pump in the IR-208 gas analyzer?
5. Arrange to collect 3 minutes of data from the 3 channels: flow, CO2, O2, and display the results on another VI, with O2 and CO2 on one chart, and flow on another. Have a waveform graph show all 180 seconds. No need to calculate tidal volume for this VI. but do convert volts from the flow meter to L/sec and convert the O2 volts to %O2 in exhaled gas.
6. Exhale shallow slow relaxed breaths (inhaling through your nose) into the mouthpiece and see how high the CO2 rises. After one minute, begin to "hyperventilation" by breathing faster and with more force. Keep up the hyperventilation for another minute. Return to normal breathing to see how CO2 returns to normal. Stop if you feel faint.
Show the results of your trial. What is the correlation between tidal volume and CO2 level? Between CO2 and O2 percentages?
Now for the tandem bike experiment:
A.V. Hill's Demonstration: Have two VI frames that toggle back and forth: one to sense breath variables for a second, the other to sense rotation for the next second.
Use reasonable caution mounting the bike seats: grab the "skyhooks" above the seats to help yourself onto a seat. let the "weak" person be on the south seat. Adjust the seat height as necessary.
Have the "strong" lab partner sit on the north seat of the reversed tandem bike; have that strong partner use a clean mouthpiece to exhale into the tubing leading to the flowmeter; it is not necessary for the whole breath to go through the flow meter--we'll be interested in the relative tidal volumes and the CO2 %. Both the "weak" and strong riders should hold onto the flexible straps above for balance, and for mounting and dismounting, and for forcing the legs to do all the work. The experiment will take 3 minutes of "riding time".
The north person will pedal forward and the other resist while (her) pedals go backward. The one going forward should try to rotate pedals at 30/min, as instructed by the person monitoring the bike "speed". However, if the "weak" pedaler can, she should try to hold the strong person to as low as 10 cycles per minute. The forward (strong) subject will breath in the monitors for volume, O2 and CO2 for 3-4 minutes. After 3 minutes the timer person will say to the "weak" person, "Pedal as hard as you can!" and we will see of she can reverse the pedals and go forward. Otherwise report how CO2 changed during the 4 minutes of sampling.
The next day the "weak" person may report soreness in her quads because she had been doing "negatives", the only way to build muscle mass in a workout.
Team "FTQ": What can you find out from Bio80 notes or from the Internet about the normal range of exhaled CO2 percent in adult humans? Is that the percentage an anesthesiologist keeps a paralyzed patient at? What is the percentage of oxygen in the atmosphere at 4000m = 12000 ft elevation, compared to sea level?
-------------------------------------
--------------------------
archive NOTES on Velocity-Tension

"At a soirée of the Royal Society conducted by Hill in 1952 two
subjects mounted a specially built tandem stationary bicycle. While one subject
pedaled the other subject resisted him through directly coupled pedals. At a
speed of 35 revolutions per minute, the subject pedaling forwards used 3.7 times
as much oxygen as the one pedaling backwards. It was demonstrated that a small
woman, pedaling backwards, could rapidly exhaust a large man pedaling forwards
against her resistance. Since oxygen consumption and heat production are closely
tied, the muscles of the woman were presumably generating far less heat, even
though the coupling of the pedals guarantees that the forces and displacements
experienced by the woman's feet were the same as those for the man's."
Thomas A. McMahon, from chapter 2 of Muscles, Reflexes and Locomotion, Princeton
Univ Press (1984).
Background:
The lecture notes tell you about Hill's equation for muscle
velocity-tension
![]()
with the figure below from McMahon's Muscles Reflexes and Locomotion
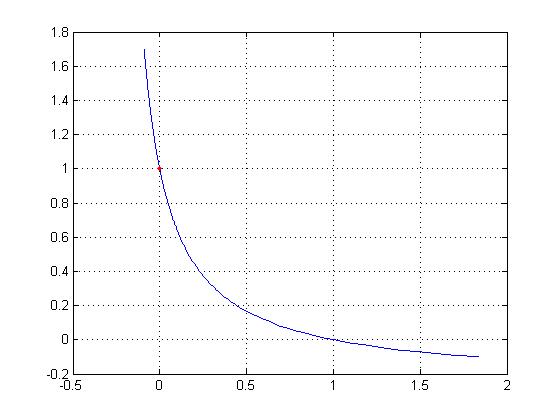
note the greater tension/fiber for negative velocity. The graph has been normalized
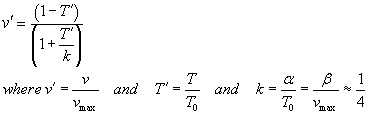
where we use 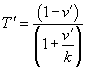 in the Matlab code described below, and you have control of constant k.
in the Matlab code described below, and you have control of constant k.
Note that α and β have the units of tension and velocity, respectively.
Muscle details:
1. Recruitment. To increase
tension (or velocity of contraction) muscle fibers are recruited, from the weakest
cells with the most endurance up to the strongest fibers, which are most easily
fatigued. Yes is it true that individual fibers can be stimulated at increasing
rates by their motoneuron axons, up to tetanus tension, but once one fiber is
pulling to the max, another fiber is recruited as necessary.
2. Velocity-dependent tension. Hill's equation shows the hyperbolic relationship
between muscle velocity and muscle tension. Note that when an active muscle is pulled
by an external load in the opposite direction of its intended contraction the muscle
is "doing a negative" (as weightlifters say) and its velocity is less
than zero. The tension per fiber is much greater in muscle fibers doing negatives
than tension in a fiber going the same speed in the opposite (positive) direction.
3. Muscle fatigue. The
FG (fast glycolytic) muscle fibers (white meat) that can exert the greatest
maximum tension are the most easily fatigued.