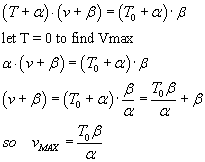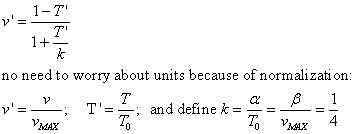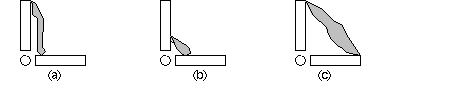Actin-myosin crossbridge cycling in skeletal
muscle
(and Hill's equation, and two demos...)
----------------
Terminology: By "muscle" we mean whole
muscle, such as the gastrocnemius or biceps or deltoid muscle.
Whole muscle is made of many muscle fibers in parallel. One fiber may be as long
as the whole muscle, but only about 10 microns in diameter. We will discuss the
orientation of muscle fibers in whole muscle-pennate muscle has diagonally oriented
fibers to increase strength at the expense of maximum length change. A muscle fiber
is a muscle cell. During development many myoblasts coalesce into
one fiber, resulting in one cell with many nuclei.
Reading
T. A. McMahon, Muscles
Reflexes and Locomotion,
Princeton Univ. Press (1984). see Chpt 1.
NOTE. Tom McMahon, a famous biomechanics professor at Harvard, inventor
of the tuned track, died in early 1999, at age of 55.
Mechanical use of muscle
Passive muscle has the length-tension properties of a nonlinear spring. Active muscle
contracts, or creates isometric tension. When it contracts (shortens) it may reduce
the angle of a joint, or force blood out of the heart, or rotate the eyes, or generate
speech. In the skeletal system muscle is fundamentally a torque or moment generator.
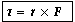
If the load a muscle pulls against is at an equilibrium torque value T0, muscle
(+tendon) won't contract, but will develop tension (isometric). How long active
muscle can maintain tension depends on how well supplied with energy it is; muscle
which develops tension most quickly is the type of muscle most likely to fatigue
during sustained activity.
Muscle joins to bone by the series connection of tendons. Tendons
are passive collagenous tissues which cannot contract. It will turn out that various
experiments which demonstrating muscle properties utilize muscle disconnected from
any bones and held by tendon, connected to a force gauge.
History. I imagine cavemen scrutinized muscle
when they cooked it over fire. Heat-treatment of muscle in rigor mortis
further stiffens the tissue by cross-linking proteins. Scientific study of muscle
probably began with the Greeks, and Aristotle. See Martha Nussbaum's translation
of Aristotle's De Moto Animalium [On The Motion of Animals]. Aristotle
made the analogy that animals moving were like puppets, controlled by "strings"
from what centuries later would be known as nerve fibers of the central nervous
system.
Passive muscle
Passive muscle has a nonlinear stress-strain curve. The first equation in TAM,
on page 8, says that the rate of stress change as a function of strain change
in unstimulated muscle is proportional to stress.
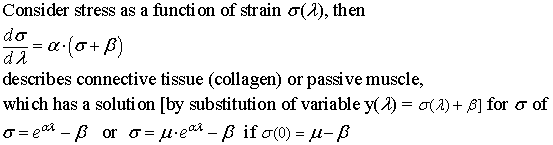
more detail:
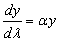
taking care to look at the limits of integration of the natural logarithm,
derived from
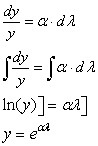
McMahon makes an analogy between stress-strain in passive muscle and stress-strain
in yarn. Pull slowly on loose yarn. As more strands of yarn are brought into
tension, the yarn resists increased pull in the nonlinear way suggested by the
equation above. In the reading of TAM
chpt 1, see Fig. 1.6. The bottom curve is for passive muscle. Below rest
length L0 muscle offers no resistance to being stretched. Loose yarn doesn't
resist until at least a few strands are pulled to their full length. Passive
stress continues out to 1.5 L0, before nearing the limit of elastic deformation.
The upper curve in TAM Fig. 1.6 shows stress-strain for active
muscle. Notice stress now starts at 0.5 L0. The curve is a combination of active
and passive effects. If the passive curve of Fig 1.6 is subtracted from the stimulated
curve, then a bell-shaped curve in Fig. 1.7, labeled"Developed" by TAM,
results. Note that the maximum tension of active muscle is not at the greatest length.
TAM explains the bell shape of the active curve later, in his chapter 3. See Fig.
3.7. At the sarcomere length where all myosin fibers can make contact with actin,
muscle exerts maximum tension. Otherwise muscle is on one side or the other of the
bell curve which represents a lack of actin-myosin crossbridge coupling. We discuss
below the crossbridge cycling mechanism which accounts for the increase in stress
of active muscle.
Actin and myosin crossbridge cycling:
Actin and myosin are filamentous proteins which interlock and overlap in a way
to produce length change and tension development in skeletal muscle. First we
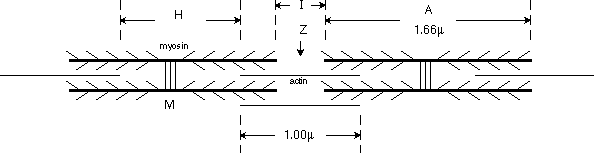
discuss the arrangement of myosin and actin in muscle sarcomeres. In
the schematic below approximate relationship between thick myosin and thin actin
fibers is illustrated. Not shown is the inhibitory tropomyosin molecule. Histological
labels are shown in capital letters. The A label marks the myosin position,
not the actin! The M line marks the midpoint of the myosin fibers,
the Z line marks the midpoint of the actin fibers. H is the
distance between the ends of actin filaments.
End-on, myosin and actin form overlapping hex and triangular arrays, as shown
below.
 www-rohan.sdsu.edu/course/ens304/public_html/section1/Muscle.htm
www-rohan.sdsu.edu/course/ens304/public_html/section1/Muscle.htm
The ratio of thin to thick filaments is 2:1
Myosin filaments are about 1.66 microns in length; actin filaments are about
1.0 microns. Bundles of myosin and actin group together in myofibrils, and the
fibrils group into muscle fibers. Remember: A fiber is one (multi-nucleated)
muscle cell.
The structure of myosin is now known in great detail, amino acid
by amino acid. In 1993 Science (24th December issue) selected myosin
as 1st runner-up for Molecule of the Year. The complete 3-D crystal structure of
myosin, to a resolution of 2.8 �, was published, and specific locations where the
myosin head could flex and where ATP could bind were suggested. A similar structure
for actin was published in 1992.
TAM chapters 3-5 go into detail about the mechanism of crossbridge
attachment and force development in muscle fibers. Here we outline what is known.
Explaining the "inverted U"
shape of muscle Length-Tension curve. See Fig 3.7, p. 67, of McMahon. 1μ
actin fiber strand on either side of 1.67μ myosin strand; eventually the
actin fibers overlap and at the depth of contraction distort myosin and reduce
tension.
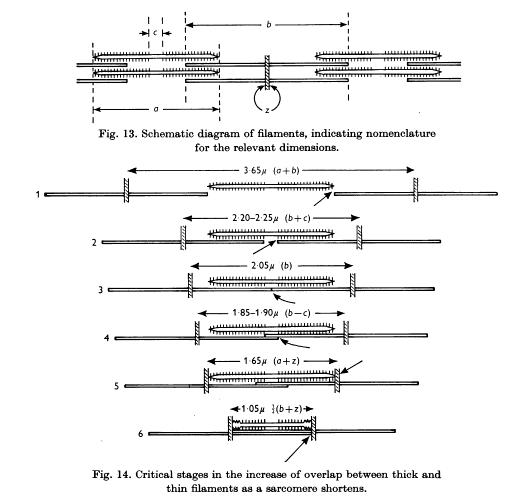
Gordon, Huxley & Julian (1966) Thick
and thin filaments overlap. Least tension for 1 and 6. Most tension 2 and 3.
EVENTS BEFORE CROSSBRIDGE CYCLING.
Before actin-myosin crossbridge cycling can occur tropomyosin must be moved out
of the way. Troponin is a component of tropomyosin; it blocks myosin from its binding
site. Tropomyosin is inhibited (moved) by calcium, which is released from sacroplasmic
reticulum in muscle. Sarcoplasmic reticulum is induced to release calcium ions
when a voltage reversal occurs at the muscle cell membrane. Neurotransmitter (acetylcholine,
ACh) from a motoneuron axon interacts with a receptor at the neuromuscular synapse
to open sodium channels which lead to the transient membrane voltage reversal. And
all that happens BEFORE crossbridges can cycle! Here is a narrative of the events:
1. Pulse travels down
motoneuron axon to neuromuscular synapse.
2. Depolarization of pulse at presynaptic terminal causes packets of ACh
to be released.
3. ACh diffuses across 10 angstrom synaptic gap to cause conformation change
in postsynaptic receptor protein.
4. Channels in receptors open and allow Na+ ions to flow in the muscle
cell.
5. Inward sodium current depolarizes muscle cell from negative potential.
6. Depolarized state causes sacroplasmic reticulum to release calcium in
muscle cell.
7. Elevated Ca++ level in muscle cell forces tropomyosin to change conformation
and pull away from actin and myosin.
As long as tropomyosin is out of the way actin and myosin
can interact.
SEQUENCE OF EVENTS WHICH OCCUR DURING CROSSBRIDGE CYCLING:
1. The head of a
myosin molecule binds to actin. There is ADP still on the myosin from a previous
cycle, and this ADP is now released, resulting in...
2. Myosin heads flex in tandem, pulling the actin strand
50-100
angstroms
into the myosin channel and contracting the muscle. If the load on the muscle
is too great, no movement occurs, but tension develops. The myosin head flex
is an example of protein conformation change. The energy required to flex the
myosin head has been previously stored in the myosin molecule and is now expended.
3. The conformation change in myosin exposes a site where ATP can bind.
If ATP is present it binds on the flexed myosin. Binding
causes release of myosin from actin.
4. The myosin head releases from the actin; Hydrolysis
of ATP to ADP causes the myosin to straighten out.
The split of ATP (into ADP + phosphate) stores energy in the myosin head
and releases some heat. Phosphate leaves the reaction site.
5. The cycle starts again if the myosin head finds a new actin binding
site.

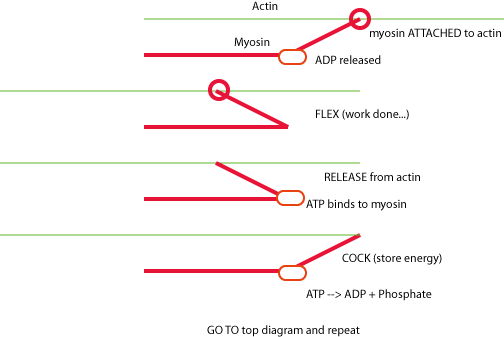
If ATP is not present and tropomyosin is inhibited, the myosin stays bound to actin. If ATP
never returns, then a state of rigor mortis ensues. If the load is such that actin doesn't move
with respect to myosin, the myosin stays linked to the actin for a longer time. The ATP
doesn't have a chance to latch onto the myosin and release its energy and heat. As a
consequence, isometric muscle releases less heat than contracting muscle. If the load is
so great that muscle is stretched during crossbridge cycling, then crossbridges are
prematurely torn apart and micro-injury occurs in the muscle. The micro-injury is a
stimulus for muscle to "repair" itself into a strengthened state. (Thus muscle-building
during weight-lifting occurs only during the "negatives." A negative occurs when the
weight is slowly lowered by the lifter. As you lower yourself during a push-up, you do a
negative. So too with a chin-up.)
Nature 352 (25 July, 1991): pages 284
& 352, "Biomechanics goes quantum," The power stroke is 10 nm; the
myosin head is about 20 nm long.
The more overlap of myosin and actin during crossbridge cycling,
the greater the tension in the muscle. Why? more myosin heads can make contact with
actin! At rest length L0 actin and myosin overlap to a maximum degree, thus the
peak in the active length-tension curve seen in TAM Fig. 1.7.
Overall muscle strength depends on length of the muscle. Try sensing
strength of grip with various wrist angles. At what wrist angle is grip strongest?
Why?
Twitch and tetanus
of muscle
Hill's equation
Years before the protein constituents of muscle were known, experiments were done
on the mechanical properties of isolated whole muscle. On page 10 of TAM, dynamic
("quick-release") experiments on isolated muscle described. A muscle is
stretched to a constant length while stimulated to tetanus, then it is released
against a variable load. Below are shown a series of quick release length changes
as suggested by the single graph of TAM's
fig. 1.8(b). The length Δx1 decreases more in the top graph because the
muscle has the opportunity to contract against the light load. As TAM says, pay
particular attention to the (negative) slope of the length change at time of release;
that's the velocity of shortening, v.
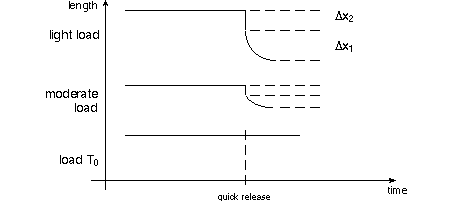
A series of quick release experiments will show an inverse relationship between
load and velocity of shortening. A. V. Hill fitted the data empirically to the equation
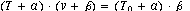
where v is shortening velocity, T is the variable load and T0 is the particular
load at which the the muscle doesn't change length, and a and b are empirical constants.
A curve of v vs T has a hyperbolic shape. (What are the asymptotes
of the hyperbola?) It turns out that a wide variety of muscles tested by quick
release result in about the same values of a and b, and that
is the importance of Hill's equation.
When velocity v is zero then T = T0.
Maximum velocity of shortening occurs when T=0.
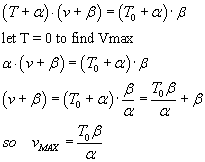
TAM says on p. 24, "For purposes of comparing different muscles, Hill's
equation can be written in a normalized form,"
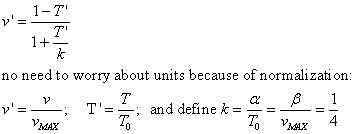
Muscle power
Power is energy/sec. Energy is force X distance.
In Hill's equation the term T�v has units of power.
Here's one way to isolate the power T�v in terms of v:
Divide both sides of Hill's equation by

which is equation 1.5 in TAM with all the steps shown.
See that the graph of Power vs Velocity has a maximum (fig in TAM). Given typical
values of a and b, maximum power is delivered when velocity
of shortening is about half of
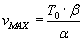
Look at the plot of T vs v of Hill's equation if v becomes negative. The force
continues to increase. What does negative velocity mean? What is a "negative"
in muscle building? It means the muscle is being stretched during stimulation,
instead of being able to contract.
Muscle mechanics around joints
Why is muscle so long? Why isn't muscle short and stubby, near the joint (b)? On
the other hand, why is muscle so short? Why not be longer, and stretch from one
end to the other? (c) What mechanical advantage (or disadvantage) occurs for the
way muscle acts to rotate bones at joints? Keep in mind that muscle has a limited
range of contraction, compared to the large amount of movement required at the ends
of the limbs.
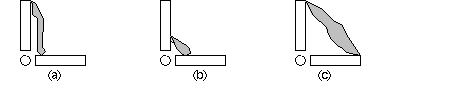
The situation in (a) means that a long muscle has a chance for a significant shortening
l�l, whereas (b) is too short to create much rotation. The great leverage of (c)
is impossible unless a pulley re-directs force, or the muscle extends across webbing.
Types of striated muscle
So far we have been assuming that striated muscle is of one kind. Yes, all striated
muscles use the same actin-myosin ratcheting mechanism for contraction and development
of tension, but in fact there are two major types of striated muscle fiber. The
distinction is greatest in non-mammalian vertebrates, where the fibers are called
FAST and SLOW. On a fiber-by-fiber basis FAST and SLOW compare in the following
ways:
| FAST |
SLOW |
| large maximum tension |
small maximum tension |
| short delay from stimulus to tension |
longer delay |
| easily fatigued |
greater endurance |
| white appearance |
the "dark meat" |
| anaerobic metabolism |
aerobic |
Swordfish fillet demo
In mammals the distinction is between fast and slow twitch muscle,
which are more properly called FG, for fast-glycolytic and SO for slow-oxidative.
Muscle fibers remain segregated in regard to which motoneurons innervate them.
By virtue of their performance as strong, easily fatigued fibers the FG fibers
are used for jumping, kicking and other movements which are intense and brief
(predator, like swordfish, going after prey...). On the other hand SO fibers
may be involved in posture and sustained activity such migratory bird flight,
where endurance is at a premium.
Muscle fatigue, strengthening and injury
Muscle changes. Over the short term it contracts and warms up. If exercised too
much, it can fatigue. Certainly white fast-twitch muscle can fatigue biochemically
after a dozen twitches because ATP is depleted. Another kind of "fatigue"
results from the micro-injury of active muscle that is lengthened; this kind of
fatigue results, over a day or so, in strengthening. Just what is the biochemical
stimulus for strengthening due to the micro-injury of "negatives"? It's
not clear to researchers. See Chpt 2 of TAM for some thoughts. It is known that
increase in muscle bulk comes from a thickening of existing fibers, instead of growth
of new fibers.
If muscle is not used, it atrophies. If muscle is in weightlessness, it atrophies a lot! Unused
muscle around bone which has healed from a fracture is smaller than the muscle around
active bone. Soviet cosmonauts learned the hard way about the atrophy of disuse; they
were unable to walk away from their space vehicle when it returned to earth after months of
weightless orbit! American astronauts exercise on treadmills in the Space Shuttle while it is
in orbit, to minimize the atrophy of muscle not required to resist gravity.
As far as I know, tendon and ligament cannot be strengthened by
use or weakened by disuse. Bone? I think studies have shown that limb bone unused
for long periods of time loses some of it capacity to "remodel" after
stress.
A muscle is thickest in its middle, thinning as it approaches
the tendons. Since, for a muscle in static equilibrium, the tension is uniform
along its length, the thin sections of muscle bear the greatest stress. The
points of contact with tendon are therefore the most likely places for injury.
You'd think Nature would have made the injury threshold of muscle greater than
it is possible for an animal to exert, so self-inflicted injury could not occur.
However, it is sometimes possible to tear or "pull" muscle by exerting
too much if the muscle has not been "warmed up." Some muscles, such
as the hamstring, are especially susceptible. If I dive cold into the pool and
start a strenuous breaststroke frog kick immediately, I could pull a hamstring.
Muscle injuries most often occur when an external force [often
contact with the ground] causes an unnatural pull. Muscles, however, are not the
weakest links in the musculo-skeletal system. As sports medicine specialists will
testify, ligaments tear before muscles pull. Thin tendons are generally more susceptible
than muscle itself, too.
Muscle can have problems other than fatigue. Cramps occur when
an errant hormonal or neural signal causes all the fibers in a muscle to contract
at once. An epileptic seizure starting in the cortex can send synchronous excitation
down through the motor system, contracting muscles simultaneously.
%---------------archive Notes-----------------------

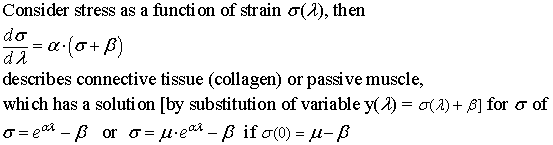
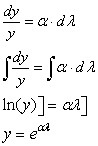

 www-rohan.sdsu.edu/course/ens304/public_html/section1/Muscle.htm
www-rohan.sdsu.edu/course/ens304/public_html/section1/Muscle.htm