Background:
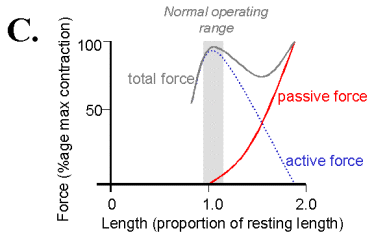
The passive elastic properties of muscles can be modelled as nonlinear springs. Their force-tension curve becomes exponentially steeper at longer lengths of stretch.
In addition to the passive force when the muscle is stimulated it contracts to produce an active force. This active force is derived from interactions of the actin and myosin filaments. The active force length tension curve is described by Sliding Filament model and has its maximum at the muscle's normal resting length in the body. At this length there is the most interaction between the actin and myosin filaments accounting for the largest active force production. At longer lengths the filaments have less opportunity for forming cross-bridges and therefore produce less force. A the muscle deceases in length past its resting length the filaments become more closely packed and are able to form fewer cross-bridges.

relates to Hooke's Law, but nonlinear.
assume muscle can be modelled as a nonlinear spring.
gastrocnemius
or sartorius muscle
Requirements:
(1) Dissection for sartorius
muscle and sciatic nerve.
We'll use Rana Pipiens, the leopard frog. Pithing, exposure of sciatic
nerve
TA or JD will do dissection (unless a premed would like to try...)
Pithing is used to euthanize the frogs by scrambling the CNS. A sharp, pointed probe is inserted through the skin into the foramen magnum between the base of the skull and the top of the vertebrae. (midline at the level of the caudal typanic oval).
Once the CNS has been scrambled, the spinal cord should be destroyed to suppress any reflexes that might disturb the experiment. The technique should be attempted only after acquiring knowledge of anatomy using skeletons, and after a period of training including practice on dead animals. This method could activate pain receptors if the proper regions of the cord are not completely destroyed.
Incisions, surgery to expose sciatic nerve, and relate to stimulator.
frog, dissection tray, gloves, instruments to use: foreceps, tweezers, pins,
glass probe, scissors, Ringer's solution, 2-0 silk thread.
<reference captured image>
use of Ringer's solution for
exposed tissue.
Description:
Also known as Ringer’s irrigation: A solution of recently boiled distilled
water containing 8.6 gm sodium chloride, 0.3 gm potassium chloride, and 0,33
gram calcium chloride per litre – the same concentrations as their occurrence
in body fluids. Ingredients are: NaCL 6 g, KCL 0,075 g, CaCl2 0,1 g, NaHCO3
0,1 g.
specifically for amphibians:
6.5 g NaCl
0.42 g KCl
0.25 g CaCl2
1 liter dH2O
Ringer experimented with various solutions containing the chlorides of sodium,
potassium, calcium and magnesium, in order to obtain a suitable physiological
saline solution which would keep the heart beating outside of the body. Several
different formulas are now used. Variants are called Locke's or Tyrode's solution.
Locke's, or Ringer-Locke solution, containins more glucose and sodium, and is
particularly used for mammals.
Set-Up:
Securely pin down the frog just above the knee, no part of the preperation should move including the dissection pan. Attach the thread tied to the Achilles tendon to the force transducer using an S hook. Adjust the length between the force transducer and the frog muscle prepartation in the tray so that it is just zero as the thread is just less than taut (your first active tension reading should be 0). Attach the stimulating electrodes to a Narishige micromanipulator stand and adjust them so that the sciatic nerve is elevated out of body and resting on the end of the electrode.
Make all the connections between the muscle preparation and the stimulator,
oscilloscope and computer. The stimulating electrodes should be connected the
stimulator. Set the stimulator to a frequency of 0.2 Hz, start with manual stimulation
by setting the output mode switch to the single position and use the current
option, (not the voltage). To find the excitation threshold set the current
to 0 and trigger the stimulator and look for a twitch, if you have it hooked
up you should be able to see it on the oscilloscope, slowly increase the current
until you have reached the maximum twitch tension, then increase the current
by about 10% to ensure a supra-maximal stimulus. You can start with the oscilloscope
at a time base of 1 msec/div and vertically 0.1-0.01 Volts/div. Trigger set
on the external input and the triggering level control to adjust to trigger
the oscilloscope sweep when the stimulator is activated. You may also want an
electrode to record the action potentials in the nerve and/or muscle (to see
contraction freq/tetanus) these should be attached to the oscilloscope and computer
through a pre-amp. The pre-amp should have the input in differential mode with
a gain of either 100 or 1000; if possible include band pass filters (10 Hz low
pass and 3 kHz high pass). The force transducer should also be hooked up to
the computer so that you can record the forces at the various lengths.
exposure of sciatic nerve, testing
(2) Measure length tension
of frog gastrocnemius (calf) or sartorius (thigh)muscle
Use of Force transducer: specs, springs.
Create LabVIEW vi to capture triggered waveforms and inspect for maximum and
steady steady state (passive) data.
to see data and generate curves.
measuring length: Narishige micromanipulator to stretch muscle.
attachment to muscle by silk thread.
mechanical deviation of force transducer?
Explore use of the stimulator (single pulse) to cause maximal twitch; record
twitch on 'scope and pick off maximum and steady state (passive) data points.
Show the passive and active L-T curves from a short muscle length when tension is zero to a "maximum" length when tension is about 80-100 grams. How reproducible is the curve?
Frog Physiology
Respiration, regulate temperature, muscle
fiber types. fast, slow
The short fibers gastrocnemius and a greater amount of connective tissue do
not show a local maximum in the total length tension curve. In comparison, the
longer, parallel muscle fibers of the sartorius contribute less to the passive
tension and therefore show a local maximum in the total length tension curve.
Background on thermocouples.
References pages 8-11 from McMahon
Links