VOLUME 49 NUMBER 4 OCTOBER 2010
Printable (PDF) Version of this issue
Effectiveness of Perforated Plexiglass Dividers as Social Grooming Devices Between Neighboring, Individually Housed Adult Male Macaca fascicularis, by L. M. Watson......1
A Case Study of Infanticide After Resident Male Replacement in Semnopithecus entellus Around Jodhpur (India), by G. Sharma, C. Ram, and, L. S. Rajpurohit......6
News, Information, and Announcements
Meeting Announcements......5
Grants Available: Fyssen Foundation Postdoctoral Research Grants......11
News Briefs......12
. . .
Barbara Orlans, Founding President of SCAW; Monkeys Use Tree Branches to Escape Researchers; Wide-Eyed Primate Photographed for First Time; Mexico Arrests Man with 18 Monkeys; New Director at Wisconsin Primate Center; National Primate Liberation Week
ASP Conservation Small Grants Awarded......13
Laboratory Equipment for Sale......14
Primatology Lectures in New York City......14
Resources Wanted and Available......15
. . .
Updated Guidance on Lab Animal Protocol Review; New Enrichment and Social Housing Resources; Database for Sharing Aging Research Models; Rhesus Photographs; Total E-Clips
Information Requested or Available: Interesting Websites......15
Announcements from Publications � JNBH......16
Three-Year Residency Program � Buffalo, New York......16
Research! by J. Schrier......16
Departments
Recent Books and Articles......17
* * *
Effectiveness of Perforated Plexiglass Dividers as Social Grooming Devices Between Neighboring, Individually Housed Adult Male Macaca fascicularis
Lyna M. Watson
Pfizer (formerly Wyeth), Comparative Medicine Department, Andover, Massachusetts
Introduction
Efforts to provide social housing to laboratory-living nonhuman primates, based on the stipulations of the Animal Welfare Act of 1987, have included a wide array of combinations with varying success across numerous species. Among these are: a semi-detached exercise cage for heterosexual vervet monkey pairs (Cercopithecus aethiops: Seier & de Lange, 1996); cross-aged, isosexual pairs of rhesus (Macaca mulatta: Reinhardt, et al., 1989) and of cynomolgus (M. fascicularis: personal observations) macaques; continuous pairing of cynomolgus macaques (Watson, 2002); limited contact through perforated panels (M. mulatta: Baker et al., 2008); and modifying caging with grooming-contact bars to foster restricted social interactions in both macaques (Crockett & Bowden, 1994; Crockett et al., 1997; 2001) and baboons (Crockett et al., 2001).
At this Pfizer (formerly Wyeth) site, there were several adult cynomolgus males which were not able to be housed in pairs. This was due to incompatibility between the animals. If an animal cannot be paired after three attempts, it is deemed socially unsuitable. We hoped that at least some of these socially unsuitable adult males could have limited or restricted social contact with a neighbor. The grooming-contact bars described by Crockett & Bowden (1994) and Crockett et al. (1997) appeared to be the best idea. However, due to staff, caging, and resource limitations, a modification of the grooming-contact bars had to be devised. The quad caging system (two single cages on top, two below; built by Lab Products, Seaford, Delaware) utilized at this site could not be altered with contact-type bars. Instead, we devised a replacement for the mesh dividers (see Figure 1) between neighboring cages. A clear, perforated, plexiglass divider was installed between neighboring cynos to test whether it altered their proximity to one another, or prompted any behavioral changes. This limited-contact divider was devised along the lines of one constructed by Baker et al. (2008 and personal communication).
Subjects: The subjects were eight wild-caught adult (aged four to six years) male cynomolgus monkeys. All the monkeys had been at this facility at least one year and were deemed unsuitable for pair-housing (failed in three different pairing attempts). The subjects were divided into four pairs of side-by-side neighbors. They were housed in quad single cages next to one another for at least three months prior to starting this study. All the monkeys were housed in standard-sized, individual stain-less steel primate cages (6.2 ft3 per cage), which were equipped with perches. The monkeys had ad libitum access to water and were provided with nine Harlan Primate biscuits/day. Fresh fruit was also given daily. Standard enrichment allocated per individual animal is a perch and two floor devices, one soft (e.g., Kong©) and one hard (e.g., Nylabone©), plus a hanging stainless steel mirror attached at the front of each cage. None of the animals were assigned to another protocol during our study. None of the subject monkeys exhibited any aberrant behaviors; thus none were assigned to special enrichment.
Device: As mentioned above, we could not modify the present caging system to install contact bars. However, because the quad caging does contain a mesh divider between each pair of cages, a comparably sized clear plexiglass divider could be modified and used as a replacement divider (see Figures 1a, 1b). Prior to starting data collection, during physical exams, measurements were made of proposed subject animals� heights, distances from shoulders to rump areas, and finger diameters. These measurements were used to map out several locations for holes in the plexiglass. These holes were located at what we anticipated to be generally used areas � near perches or along the top sides of dividers. Hole diameter size was restricted so that none allowed an animal to go as far as its wrist. For the dimensions of the holes themselves, we measured across knuckles, across hand (from base of thumb across back of hand), and circumference of the wrist, averaging 7.5 cm, 9.1 cm, and 8.5 cm, respectively. We therefore made round holes 9 cm in diameter. The spaces between pairs of cages, holding either mesh or plexiglass dividers, had the same dimensions: 29� wide x 29� long x 2� deep; the plexiglass dividers were �� thick.

Figure 1a: A mesh divider, 29 x 29 inches wide. Mesh on the left is .5-inch squares; that on the right, .75 x .5 inch rectangles.
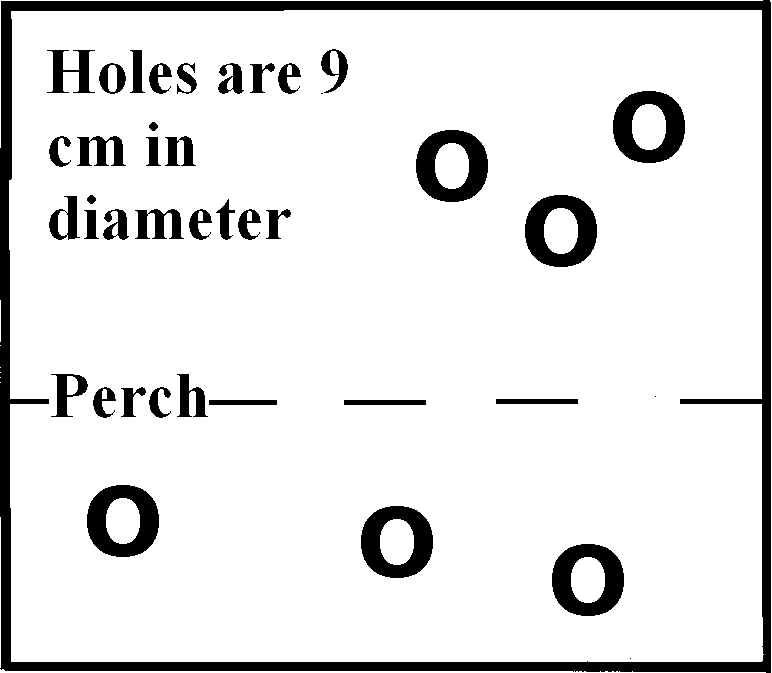
Figure 1b: A clear plexiglass divider, also 29� x 29�. The holes are 9 cm in diameter. The perch is 12� from the bottom.
Data Collection: Behaviors: Thirty-one different behaviors were recorded (see Table 1). Data were collected twice a day, three days a week for three weeks with mesh dividers in place, and then for three weeks with plexiglass dividers in place. The scan samples were five minutes long with twelve-second intervals; that is, 25 scans/session per animal (50 scans/day x 3 days/week = 150 scans/week/ animal). This equals a total of 450 scans/subject with mesh divider and 450 scans/subject with plexiglass divider. A cross-observer reliability check resulted in 95% agreement.
|
• Touching o Divider o Other part of cage o Neighbor • Scratching • Grooming o Self o Neighbor • Play o By self o With other o With enrichment device • Lipsmack • Yawn • Aggression o Grab o Slap o Poke o Open mouth |
o Eyebrows raised • Shake bar • Look o Away o At researcher or other person in room o At television o At other monkey o at neighbor • Locomotion o Normal o Stereotypic • Sit passively • Vocalize • Eat • Forage • Drink • Submissive behavior (e.g., tail down, shoulders crouched) • Pulling own hair |
Table 1: Behaviors recorded.
Data Analysis: For each behavior or set of behaviors, the number of scans with the plexiglass divider in place was compared to the number with the mesh divider, using a negative binomial regression model and generalized estimating equations (GEE) to account for the repeated measures nature of the data.
Analysis of the proximity data began with calculation of a �mean� proximity for each animal for each divider type. The mean proximity was computed as the average of the nominal proximity values (1 = at divider; 2 = within 6�; 3 = 7-12� away; and 4 = beyond 12�) weighted by the number of scans at each distance. Mean proximity with the plexiglass divider in place was compared to that with the mesh divider in place, using a two-tailed paired t-test and Wilcoxon signed-rank test. Further analysis was performed using Spotfire.
Of the thirty-one behaviors scored during each session, seven resulted in statistical significance at the 0.05 level. They are: Look Away, Look at Other Monkey, Look at Neighbor Monkey, Look at Person, Look at Television, Non-stereotypic Locomotion, and Play with Enrichment Device. Four behaviors did not occur at all, or so little that they did not register frequently enough to do a statistical analysis. Those were: Stereotypic Locomotion, Submissive Behavior, Groom Neighbor, and Open Mouth (in the Aggression set). This left 27 behaviors remaining for statistical analysis. Behaviors of a similar nature were analyzed individually, as well as collapsed and analyzed under one term, i.e., Play or Locomotive Behaviors. These specific results are provided in Tables 2�5.
Proximity: A monkey�s proximity to either the mesh or the plexiglass divider was measured using a scale of �majority of animal in place�. That is, if most of the animal�s body was located within 7� of the divider, that score is given. This was at the discretion of the observer since no tape or other means of dividing the cage was in place. There was not a statistically significant difference in the monkeys� proximity to the plexiglass divider compared to the mesh one (paired t-test: p = .94).
Despite this nonsignificant outcome, a noteworthy pattern was discerned (see Table 2). The actual frequency outcomes for the animals� proximities to one another are presented. Three of the eight monkeys displayed a shift in location preference to their neighbor depending on whether the mesh or the plexiglass was present. The most dramatic shift occurred in Pair I in which Monkey #1 completely shifted his proximity to his neighbor, from often being furthest away from the mesh divider to mostly being right next to the plexiglass when it was in place. This animal and monkey #8 in Pair IV, with a slightly less dramatic shift (from Beyond 12� to Within 6�), were the only monkeys to display such an impressive shift in location to the divider or to his neighbor depending on which type of divider was present.
| Pair I | Pair II | Pair III | Pair IV | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Monkey 1 | Monkey 2 | Monkey 3 | Monkey 4 | Monkey 5 | Monkey 6 | Monkey 7 | Monkey 8 | |||||||||
| Proximity: | Mesh | Plex | Mesh | Plex | Mesh | Plex | Mesh | Plex | Mesh | Plex | Mesh | Plex | Mesh | Plex | Mesh | Plex |
| At Divider | 100 | 345 | 226 | 43 | 364 | 438 | 88 | 42 | 429 | 240 | 20 | 48 | 447 | 284 | 25 | 88 |
| Within 6� | 41 | 45 | 211 | 249 | 37 | 4 | 36 | 32 | 21 | 24 | 7 | 4 | 1 | 14 | 14 | 130 |
| 7-12� Away | 33 | 20 | 11 | 108 | 16 | 6 | 148 | 80 | 0 | 34 | 9 | 0 | 2 | 10 | 3 | 108 |
| Beyond 12� | 276 | 40 | 2 | 50 | 25 | 2 | 178 | 296 | 0 | 152 | 414 | 398 | 0 | 142 | 408 | 115 |
| Mean Distance | 3.1 | 1.5 | 1.5 | 2.4 | 1.3 | 1.0 | 2.9 | 3.4 | 1.0 | 2.2 | 3.8 | 3.7 | 1.0 | 2.0 | 3.8 | 2.6 |
| Paired t-test p value = 0.94 | ||||||||||||||||
Table 2: Monkeys� proximities to either mesh or solid plexiglass dividers: number of scans at each distance.
| Pair I | Pair II | Pair III | Pair IV | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Monkey 1 | Monkey 2 | Monkey 3 | Monkey 4 | Monkey 5 | Monkey 6 | Monkey 7 | Monkey 8 | |||||||||
| Mesh | Plex | Mesh | Plex | Mesh | Plex | Mesh | Plex | Mesh | Plex | Mesh | Plex | Mesh | Plex | Mesh | Plex | |
| Look Away | 1 | 0 | 18 | 5 | 16 | 8 | 20 | 16 | 5 | 10 | 10 | 5 | 7 | 1 | 45 | 27 |
| p value = 0.0003 | ||||||||||||||||
| Look at Person | 36 | 76 | 62 | 86 | 41 | 50 | 104 | 128 | 74 | 101 | 138 | 152 | 28 | 58 | 49 | 63 |
| p value = .09 | ||||||||||||||||
| Look at Television | 24 | 3 | 87 | 44 | 102 | 63 | 68 | 42 | 29 | 52 | 35 | 39 | 25 | 25 | 42 | 30 |
| p value = .06 | ||||||||||||||||
| Look at Other Monkey(s) | 6 | 2 | 20 | 4 | 5 | 0 | 8 | 0 | 6 | 2 | 15 | 7 | 5 | 2 | 3 | 5 |
| p value = .017 | ||||||||||||||||
| Look at Neighbor Monkey | 5 | 6 | 24 | 57 | 27 | 35 | 44 | 46 | 34 | 46 | 42 | 71 | 44 | 64 | 78 | 108 |
| p value = .0084 | ||||||||||||||||
Table 3: Monkeys� looking behaviors with either mesh or solid plexiglass dividers: number of scans.
Visual or Looking Behaviors: Visual behavior, or Looking, was scored and placed into one or more of five specific behaviors. They were: Look Away, Look at Neighbor (neighboring individual on other side of divider), Look at Other Monkey (not Neighbor), Look at Person, or Look at Television. A majority of the subjects exhibited three of these specific looking behaviors more frequently when the mesh, rather than the plexiglass, was in place. Those behaviors were Look Away, Look at Other (non-neighbor) monkey, and Look at Television. The behavior of Look Away was displayed by six of the eight subjects more frequently when the mesh, rather than the plexiglass, was in place (p< .0003). That is, the animals were more likely to glance or look away (avoid direct eye contact) from their neighbor or the human when the mesh, rather than the plexiglass, was in place. Looking at Other Monkey refers to the subject looking at another animal in the room. This specific behavior was displayed more frequently (p < .017) when the mesh, rather than the plexiglass, was in place. The subjects� tendency to watch the television more frequently when the mesh was in place was marginally statistically insignificant (p < .06) (see Table 3).
The remaining two statistically significant Looking Behaviors were Looking at Neighbor (p < .008) and Looking at Person (p < .09). In contrast to the above Looking Behaviors, in these two specific behaviors the subject monkeys displayed higher tendencies when the plexiglass, rather than the mesh, was in place. Unlike any other Looking Behavior, in these two specific conditions (looking at neighboring monkey and humans in room), all the subjects did so when clear plexiglass, rather than mesh, was in place. Possibly, it was much easier for them to see a lot more of their surroundings when the plexiglass, rather than the metal mesh, was in place. With the mesh in place, an object may not be as distinct.
Locomotive Behaviors: The next statistically significant behavior is �Non-stereotypic Locomotion� at p <.0018. Seven of the eight subjects displayed a higher frequency of this behavior with the mesh divider in place than with the plexiglass (see Table 4). That is, they were less likely to move about when the plexiglass was present. This tendency is reflected in Table 2�s pattern, based on mean �distance� outcome.
| Pair I | Pair II | Pair III | Pair IV | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Monkey 1 | Monkey 2 | Monkey 3 | Monkey 4 | Monkey 5 | Monkey 6 | Monkey 7 | Monkey 8 | |||||||||
| Mesh | Plex | Mesh | Plex | Mesh | Plex | Mesh | Plex | Mesh | Plex | Mesh | Plex | Mesh | Plex | Mesh | Plex | |
| 4 | 10 | 20 | 5 | 18 | 10 | 38 | 10 | 22 | 10 | 24 | 6 | 24 | 6 | 48 | 38 | |
| p value = 0.0018 | ||||||||||||||||
Table 4: Monkeys� locomotive behavior with either mesh or solid plexiglass dividers: number of scans.
Additional Behaviors: None of the other measured behaviors resulted in statistical significance across conditions (mesh vs. plexiglass divider). However, one of the patterns discerned and worth mentioning is their playing with the enrichment devices, which resulted in borderline statistical significance (p < .07). Seven of the eight monkeys (monkey #8 was the exception) displayed more play behavior when mesh, rather than plexiglass, was in place. The frequencies of this behavior are presented in Table 5.
| Pair I | Pair II | Pair III | Pair IV | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Monkey 1 | Monkey 2 | Monkey 3 | Monkey 4 | Monkey 5 | Monkey 6 | Monkey 7 | Monkey 8 | |||||||||
| Mesh | Plex | Mesh | Plex | Mesh | Plex | Mesh | Plex | Mesh | Plex | Mesh | Plex | Mesh | Plex | Mesh | Plex | |
| 105 | 21 | 24 | 10 | 67 | 41 | 10 | 7 | 46 | 42 | 35 | 0 6 | 0 | 15 | 44 | ||
| p value = 0.0547 | ||||||||||||||||
Table 5: Monkeys� play with enrichment device with either mesh or solid plexiglass dividers: number of scans.
Adult male cynomolgus monkeys tend not to spend a large amount of time in close proximity to one another in the wild, nor in other settings (van Noordwijk & van Schaik, 1985). This lack of close proximity was reflected in the outcome of the present investigation, regardless of whether the mesh or the plexiglass divider was in place. Although several pairs showed some changes in relative location in their cages when the mesh vs. the plexiglass was present, overall there was not a significant tendency to be closer to each other with the plexiglass. Further, the presence of a different type of divider, one equipped hopefully to encourage grooming, not only did not encourage them to get closer, but definitely did not result in increases in mutual grooming. Baker et al. (2008) determined that grooming partitions with holes (2� circumference) were not productive for rhesus males housed next to one another. Crockett et al. (1997) found that grooming partitions were not as effective or used as frequently by rhesus male-male neighbors as by female-female neighbors or male-female neighbors.
In the latter two studies a pair had to be separated and removed from the study due to injury. In the present study, shortly after the observation period ended, one monkey was bitten on the mouth and tongue by his neighbor while the plexiglass was in place. Our veterinarian instructed that the remaining plexiglass dividers be permanently removed and replaced with the original mesh dividers. The monkeys� relatively infrequent use of plexi-glass with holes for grooming, coupled with the likelihood of injuries occurring, leads this researcher not to be inclined to recommend them as an alternative to social housing, at least for adult male cynomolgus macaques.
The author wishes to thank Dr. Mel Balk and the Comparative Medicine staff at Pfizer (formerly Wyeth), with special thanks to Carl Gudowski for assistance in data collection. Also, thank you to Fred Immerman and Jason Machan for their statistical analysis and review.
Baker, K., Bloomsmith, M., Griffis, C., Oettinger, B., Schoof, V., Clay, A., & Maloney, M. (2008). Benefits of isosexual pairing of rhesus macaques (Macaca mulatta) vary with sex and are limited by protected contact but not by frequent separation. American Journal of Primatology, 70[S1], 44.
Bauman, V., Coke, C., Green, J., Moreau, E., Morton, D., Patterson-Kane, E., Reinhardt, A., Reinhardt, V., & Van Loo, P. (Eds.). (2007). Pair-housed monkeys with head cap implants. In: Making lives easier for animals in research labs (Chapter 8.9). Washington, DC: Animal Welfare Institute.
Bielitzki, J., Susor, T., Elias, K., & Bowden, D. (1990). Improved cage design for single housing of social nonhuman primates. Laboratory Animal Science, 40, 428-431.
Crockett, C., Koberstein, D., & Heffernan, K. (2001). Compatibility of laboratory monkeys housed in grooming-contact cages varies by species and sex. American Journal of Primatology, 54(Suppl.), 51.
Crockett, C. M., Bellanca, R. U., Bowers, C. L., & Bowden, D. M. (1997). Grooming-contact bars provide social contact for individually caged laboratory macaques. Contemporary Topics in Laboratory Animal Science 36, 53-60.
Crockett, C., & Bowden, D. (1994). Challenging conventional wisdom for housing monkeys. Lab Animal, February, 29-33.
Seier, J. V., & de Lange, P. W. (1996). A mobile cage facilitates periodic social contact and exercise for singly caged adult vervet monkeys. Journal of Medical Primatology, 25, 64-68.
Social housing of nonhuman primates with cranial implants: A discussion (2009). Laboratory Primate Newsletter, 48[2], 1-2, <brown.edu/Research/Primate/LPN482.html#implants>.
Van Noordwijk, M., & van Schaik, C. (1985). Male migration and rank acquisition in wild long-tailed macaques (Macaca fascicularis). Animal Behaviour, 33, 849-861.
Watson, L. (2002). A successful program for same- and cross-age pair-housing adult and subadult male Macaca fascicularis. Laboratory Primate Newsletter, 41[2], 6-9, <www.brown.edu/Research/Primate/lpn41-2.html#watson>.
-------------------------------------------
Author�s address: Pfizer, Comparative Medicine Dept., Bldg G, One Burtt Rd., Andover, MA 01810 [e-mail: [email protected]].
-------------------------------------------
* * *
The 28th Annual Symposium on Nonhuman Primate Models for AIDS, hosted by the Tulane National Primate Research Center, will be held from October 19�22, 2010, in New Orleans, Louisiana. The co-chairs will be Drs Ronald S. Veazey and Preston A. Marx. See <www.tnprc.tulane.edu/pdf/2010AIDSsymposium.pdf> for details.
The Association of Professional Wildlife Educators (APWE) will be hosting a conference March 2�4, 2011, at Frank Buck Zoo in Gainesville, Texas. The theme this year is: �Creative ideas for 21st century environmental education�. See <www.apwe.org>.
* * *
A Case Study of Infanticide After Resident Male Replacement
in Semnopithecus entellus Around Jodhpur (India)
Goutam Sharma, Chena Ram and L. S. Rajpurohit
Animal Behaviour Unit, Department of Zoology, Jai Narain Vyas University
Introduction
The Hanuman langur, Semnopithecus entellus (Dufresne, 1797), is the most widely distributed of 19 nonhuman primate species found in the Indian subcontinent. It is a highly adaptable species (Roonwal & Mohnot, 1977; Wolfheim, 1983). Social units in Hanuman langurs include one-male multi-female troops, multi-male multi-female troops, and all-male bands.
The langur population around Jodhpur has been well studied during the last four decades. In this part of their habitat langurs usually form one-male multi-female troops (Mohnot, 1974). Multi-female troops are generally matrilineal, with females remaining for life in their natal troops while males emigrate, usually as juveniles, to join all-male bands. Resident males of these multi-female troops are usually replaced after two or three years (range = 3 to 60 months) (Mohnot et al., 1987; Rajpurohit, 1987; Raj-purohit & Mohnot, 1988; Sommer & Rajpurohit, 1989; Rajpurohit & Sommer, 1993). Two types of resident replacement processes have been reported: abrupt changes and gradual changes (Rajpurohit, 1987; Rajpurohit & Mohnot, 1988). During the process of an adult male replacement, the resident is driven out by invading males from an all-male band. A variety of social changes are seen in this species. Resident male change or replacement is an important social change, periodically seen in the one-male multi-female troops.
Infanticide has been observed in many mammals including several primate species (see Hrdy, 1979; Hausfater & Hrdy, 1984). Male replacement and infant killings in Hanuman langurs have been reported at several study sites in India: Dharwar (Sugiyama, 1965a), Jodhpur (Mohnot, 1971; Makwana, 1979; Sommer & Mohnot, 1985; Agoramoorthy & Mohnot, 1988; Rajpurohit & Chhangani 2003; Rajpurohit et al., 2003), Mount Abu (Hrdy, 1974), Kanha (Newton, 1986), and Tirunelveli (Ross, 1993), as well as in Nepal (Borries, 1997). It is notable that at all the sites where infanticide has been reported, the troop structure was either already predominantly one-male or after the take-over (resident male change) seemed to become functionally one-male.
It has been difficult to explain the form and function of infanticide in terms of modern evolutionary theory. Hrdy�s hypothesis that infanticide has evolved primarily to procure reproductive advantage to males has received much attention. A new resident male could speed up the mothers� sexual receptivity by eliminating young infants unlikely to be his own, so that they could then bear his offspring (Hrdy, 1974). However, questions remain regarding wounding and disappearances of older infants and juveniles at the time of take-over and after resident male change (Vogel & Loch, 1984; Agoramoorthy & Mohnot, 1988). A second hypothesis suggests that an infanticidal male might kill infants in order to increase the resources available for himself and his relatives (Rudran, 1979). Mohnot (1971) explains infanticide as arising from a spill-over of aggression during take-over.
The present paper describes the process of adult male replacement followed by infanticide in a one-male multi-female troop of langurs at Arna (B-29), near Jodhpur (Figure 1). Our observations suggest a �terror strategy� hypothesis similar to Mohnot�s: the exhibition of male power to troop members as well as to rival males.
A demographic study of Hanuman langurs, Semnopithecus entellus, at Jodhpur, Rajasthan (altitude 240 m, latitude 26� 18� N, longitude 73� 08� E), has been in progress for the last seven years. The number of langurs living in this genetically isolated population around Jodhpur, within a total area of about 150 km2, has fluctuated from about 900 or 1300 (Mohnot et al., 1987; Rajpurohit, 1987) to about 1950 individuals (Mohnot, 2000). The most recent survey and census of the Jodhpur langur population (during May�June, 2009) counted 2007 langurs in this isolated population. This population was comprised of 38 one-male multi-female troops and 15 all-male bands. Troop sizes varied from 6 to 142, averaging 47.6 individuals; while the all-male band size varied from 3 to 69, averaging 15.3. The adult male to adult female sex ratio was 1:5.6.
The habitat used by the langurs around Jodhpur includes open scrub forest, fields, farms, and orchards (Mohnot, 1974). All langur groups feed on the natural vegetation, which is xerophytic, and also raid crop fields and orchards. However, these animals are well habituated to observers because they are also provisioned by local people.
Study group: The present report deals with a one-male multi-female troop at Arna (B-29), an outlying field site of the Jodhpur study area, which is 20 km west of the city of Jodhpur (see Figure 1). This troop had 24 individuals: an adult resident male, 11 adult females, 3 juveniles (7 to 12 months old), and 9 infants of both sexes. Three of the infants were �black-coat� (birth to 3 months), and six were �white-coat� (4 to 6 months) in the second week of September, 2009, and appeared normal. One �black-coat� was changing color and another �black-coat� was born before October 1.
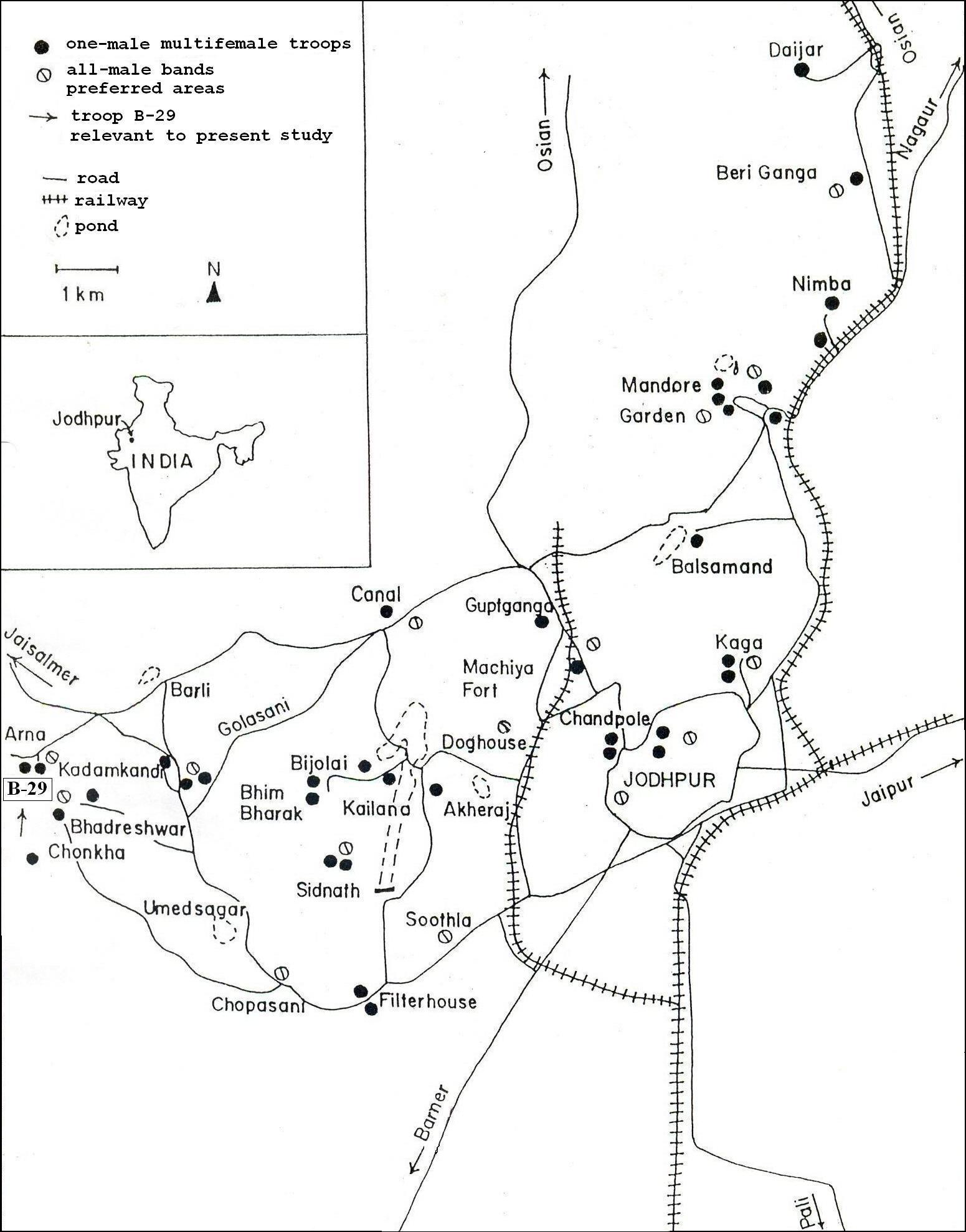
Figure 1: Location of one-male multi-female troops and all-male bands around Jodpur.
Three or four of the 17�19 males in a nearby all-male band used to approach the Arna troop occasionally, but the resident male of B-29 was capable and powerful enough to chase the males away whenever they appeared in his troop�s territory.
October 1, 2009
7:00 a.m. B-29 troop appeared normal near its resting site and there were no other adult males interacting or seen around this troop, which ranged throughout an area of about 1.2 km2.
October 4, 2009
7:25 a.m. B-29 troop was on the hillock just above the temple. We heard a teeth-grinding sound by the resident male and also observed him air-biting towards the north. Looking through binoculars in that direction, we noticed two langur males approaching the troop.
7:35 a.m. We went to the troop, which was climbing a hillock, and watched the resident male, whose left leg was severely injured. He was not able to use that leg properly. He continued teeth-grinding and air-biting toward the invaders.
The resident male�s injury might have been from fighting with the invading males one or two days before. But he still looked powerful enough and active even after this injury. He was jumping and air-biting towards the invading males, who were sitting about 200�250 m away. He even ran closer to the place where the males were sitting and chased them a bit. The invading males resisted and did not leave the place.
7:45 a.m. The alpha male came back to the troop and jumped a bit; all the infants of the troop screamed together, some of them approaching the resident male and touching his back or tail. Of the 10 infants in this troop, most were with their mothers and hidden back under the rocks. The resident male�s wound was bleeding continuously due to his running and jumping. He appeared uneasy and was air-biting towards the outside males.
7:55 a.m. The resident again went to chase the invading duo, but did not succeed and came back to his troop. Now the outsider males approached the troop. For about 8�10 minutes, there was no activity from either side.
8:15 a.m. The invading males came close, and sat again. The alpha male started teeth-grinding and jumping; the infants screamed. When the invaders came up to the troop, all the infants were retrieved by their mothers and the troop members moved a bit.
8:18 a.m. The alpha tried to chase the males away; and there was serious fighting between the resident male and the invaders. The resident male was unable to manage due to the serious injury on his leg. He fell (from about 19 m height) from the big rock of the hillock to the platform of the temple below. He came up within 10�15 seconds and started teeth-grinding and air-biting again while sitting about 15 m away.
8:25 a.m. Some females tried to chase the two invaders, but did not succeed. And so the invaders were in the troop. The alpha male was sitting just 10 meters away. The whole troop still was with the resident male.
8:35 a.m. The resident male continued teeth-grinding and looking at his wound again and again, putting his hand on it. The wound was bleeding, and he was not at all able to use that injured leg for moving or walking. He still looked aggressively at the invaders and continued to air-bite and teeth-grind. The invaders were also grinding their teeth.
8:45 a.m. One female of the troop (without any infant) chased the invaders and displaced them from the place they were sitting.
8:50 a.m. Teeth-grinding from both sides continued and other females also tried to chase the outsiders, but were chased themselves now. And the invaders stayed there.
8:52 a.m. One male �white-coat� infant went close to one of the invaders and touched him. Immediately his mother followed, retrieved him, and ran back to the troop. 8:55 a.m. Teeth-grinding continued. One female went to chase the invaders. She was not successful, but after being joined by another adult female they succeeded in chasing them a bit. However, the invading males did not harm the females.
8:58 a.m. The alpha still was teeth-grinding, which was not too harsh now. Meanwhile, females chased the males 2 or 3 times. But the outsiders resisted.
9:00 a.m. The alpha male started to feed on provisioned food (e.g., cucumber, eggplant, and nuts). Both the invading males were also observed feeding on some of the provisioned food lying just 5 m from the resident male.
9:05 a.m. Troop members moved again, and the alpha male now followed them. The outsider males were seen sitting at the same place. The troop was staying with the same adult male, who had been exhausted due to violent fighting with the invaders for the last two hours. But the troop members had full confidence in him.
October 6, 2009
7:20 a.m. The injured resident male was not seen with the Arna troop. One of the invading males was the new alpha of this troop. Neither the old resident male nor the second invading male was seen around. Now the troop had a new resident male.
October 7, 2009
7:40 a.m. The Arna troop had a new resident male; all the infants appeared normal. The displaced resident male was not found even after we searched the whole area.
On October 7 two of the black-coat infants were safe with their mothers, but the third and youngest was noted as a target of the new resident male�s attention, while his mother was keeping her distance. Then we could not visit the troop for one day.
On October 9, at 7.30 a.m., troop B-29 was found near the temple at Arna. However, we didn�t find the youngest black-coat infant. We looked around but that infant was not found, either dead or alive. Then we checked a white-coat infant and found an injury on its right buttock. That injury looked not too old, maybe a day or two. The mother was observed not allowing the infant to leave her lap. The new resident appeared very aggressive; two females presented to him, but the male showed no interest. Neither the ousted resident nor the other invading male was seen, even after we searched here and there.
On October 10, at 8.00 a.m., the new resident was observed with troop B-29, which was on the hill. We saw a dead black-coat infant near a body of water, and a female sitting close to it. This female might have been the mother of the victim, which had probably been killed by the new resident.
In November, 2009, the new resident was nearly accepted by the females of this troop. But still the injured white-coat�s mother has been seen keeping her distance from him. There have been no new births during the last two months and the troop has appeared normal. The new resident male has appeared somewhat aggressive on a few occasions, and has been seen attacking the infants in the troop again.
Replacement of a resident male is one of the most important social changes seen periodically in Hanuman langurs. Large numbers of authors have reported such replacement in langurs (Hrdy, 1977; Jay, 1962; Makwana & Advani, 1981; Mohnot, 1971; Rajpurohit, 1991; Rajpurohit & Chhangani, 1999; and Sugiyama, 1965b). Different langur populations have, however, been found to show considerable variation in the pattern of resident male change, which may be either gradual or abrupt.
In Sugiyama�s (1965b) well documented case of adult Hanuman langur male replacement in Dharwar, the usurping male drove the resident adult and immature males from the group and killed infants still dependent on their mothers. Mohnot (1971) and Hrdy (1977) described similar replacement accompanied by infanticide among Hanuman langurs at Jodhpur and Mt. Abu. Rudran (1973) reported adult male replacement in purple-faced langurs (Presbytis senex) in Sri Lanka. Although he did not report male attacks on infants, large number of infants did disappear from the group in which male changes occurred. Given all the above reports and hypotheses, as well as the present observations, we postulate that forcible adult male replacement may play a crucial role in maintaining the one-male troop structure in Hanuman langurs. In the present study, the resident male replacement took place within a period of two days. Such abrupt resident male replacement is not very common in this species.
Regarding infanticide, the sexual selection hypothesis suggests it results in increased reproductive output for the incoming male (Hrdy, 1974, 1977). Leland et al. (1984) suggest that infanticide is more likely to occur in one-male groups than in multi-male groups. In multi-male groups, paternity may be confused, leading to the possibility that more than one male may defend an infant from attack. Also, the chances of an infanticidal male siring the mother�s next infant are lower than in one-male groups. Hence the costs of infanticide are increased and the benefits decreased in multi-male groups. The infanticide attacks we observed in the Arna troop occurred in a one-male situation.
Curtin and Dolhinow (1978) suggested that infanticide is linked to an unnaturally high primate population density brought about by human disturbance. However, other studies have seen infanticide in undisturbed populations of langurs (Newton, 1986) and in other primate species (Struhsaker & Leland, 1987). Our observations offer little support for the social pathology theory. Although the Jodhpur langur population is found near human habitation, the density of langurs in the area (approximately 14.5/km2, according to the present study and Rajpurohit et al., 2003) is not high.
Rudran (1979) suggests that an infanticidal male might kill infants in order to increase the resources available for himself and his relatives. However, this hypothesis does not explain why the new male in our study attacked the youngest infant, the animal consuming the least resources, but not older infants and juveniles who consume more (Hrdy, 1977; Sommer & Mohnot, 1985; Agoramoorthy & Mohnot, 1988).
Another hypothesis explains infanticide as arising from high levels of aggression during take-over (Mohnot, 1971). This idea is not contradicted by the infanticide attacks we report here. In Mohnot�s study, the new resident male of troop B16 attacked the two youngest infants during a time of high aggression and extreme tension. He attacked at that time only; after that he appeared normal. We propose here that the function of infanticide can be explained by the exhibition of the male�s power � a �terror strategy� � to rival males nearby and to the troop members, just to let them understand his supremacy. It might be simply coincidence that more male infants are victimized, as they are slightly more in number in the infant sex ratio. The sexual selection, resource competition, or social pathology hypotheses are not supported at this study site. However, we do find support for the idea that infanticide is associated with a period of high aggression during male replacement, as proposed by Mohnot (1971).
The authors are grateful to Dr. Devendra Mohan, Head, Department of Zoology, J.N.V. University, Jodhpur, for logistic support. The fieldwork is a part of research activity conducted under a program. Thanks are due to the University Grants Commission, New Delhi, for financial support as part of a major project. Thanks also to the office staff of the project for typing this manuscript.
Agoramoorthy, G., & Mohnot, S. M. (1988). Infanticide and juvenilicide in Hanuman Langurs (Presbytis entellus) around Jodhpur, India. Human Evolution, 3, 279-296.
Bernstein, I. S. (1968). The lutong of Kuala Slangor. Behaviour, 32, 1-16.
Bishop, N. (1975). Social behaviour of langur monkeys (Presbytis entellus) in a high altitude environment. PhD thesis, University of California, Berkeley.
Hrdy, S. B. (Ed.). (1977). The langurs of Abu: Female and male strategies of reproduction. Cambridge: Harvard University Press.
Hrdy, S. B. (1979). Infanticide among animals: A review, classification and examination of the implications for the reproductive strategies of females. Ethology and Sociobiology, 1, 13-40.
Hrdy, S .B. (1974). Male-male competition and infanticide among the langurs (Presbytis entellus) of Abu, Rajasthan. Folia Primatologica, 22, 19-58.
Jay, P. (1965). The common langur of North India. In I. Devore (Ed.), Primate Behaviour (pp. 197-249). Winston, NY: Holt, Rinehart & Winston.
Leland, L., Struhsaker, T. T., & Butynski, T. M. (1984). Infanticide by adult males in three primate species of Kibale Forest, Uganda: A test of hypotheses. In G. Hausfater & S. B. Hrdy (Eds.), Infanticide: Comparative and evolutionary perspectives (pp. 151-172). Hawthorne, NY: Aldine.
Makwana, S. C. (1979). Infanticide and social change in two groups of the Hanuman langur (Presbytis entellus) at Jodhpur. Primates, 20, 293-300.
Makwana, S. C., & Advani, R. (1981). Social change in Hanuman langurs, Presbytis entellus around Jodhpur. Journal of the Bombay National Historical Society, 78, 152-154.
Mohnot, S. M. (1971). Some aspects of social changes and infant killing in the Hanuman langur, Presbytis entellus (Primates: Cercopithecidae) in Western India. Mammalia, 35, 175-198.
Mohnot, S. M. (2000). Indo-US Primate Project Annual Report, Year 06.
Mohnot, S. M. (1974). Ecology and behaviour of the common Indian langur, Presbytis entellus. PhD Thesis, University of Jodhpur.
Mohnot, S. M., Agoramoorthy, G., Rajpurohit, L. S., & Srivastava, A. (1987). Ecobehavioural studies of Hanuman langurs, Presbytis entellus. Technical Report, Department of Environment & Forest, Goi.
Mohnot, S. M., Gadgil, M., & Makwana, S. C. (1981). On the dynamics of the Hanuman langur population of Jodhpur (Rajasthan, India). Primates, 22, 182-191.
Moore, J. (1985). Demography and sociality in primates. PhD thesis, Harvard University, Cambridge.
Newton, P. (1987). The social organisation of forest Hanuman langurs, Presbytis entellus. International Journal of Primatology, 8, 199-232.
Newton, P. N. (1986). Infanticide in an undisturbed population of forest Hanuman langurs (Presbytis entellus). Animal Behaviour, 34, 785-789.
Rajpurohit, L. S., & Chhangani, A. K. (1999). Study of the process of resident male change in a unimale bisexual group of Hanuman langur, Semnopithecus entellus, around Jodhpur, Rajasthan. Cheetal, Journal of the Wildlife Preservation Society of India, 37, 13-17.
Rajpurohit, L. S., & Mohnot, S. M. (1988). Fate of ousted male residents of one-male bisexual troops of Hanuman langurs (Presbytis entellus) at Jodhpur, Rajasthan. Human Evolution, 3, 309-318.
Rajpurohit, L. S., & Sommer, V. (1993). Juvenile male emigration from natal one-male troop in Hanuman langurs. In M. E. Pereira & L. A. Fairbank (Eds.), Juvenile primates: Life history, development and behaviour (pp. 86-103). New York and Oxford: Oxford University Press.
Rajpurohit, L. S. (1991). Resident male replacement, formation of new male band and paternal behaviour in Presbytis entellus. Folia Primatologica, 57, 159-164.
Rajpurohit, L. S. (1987). Male social organization of Hanuman langurs (Presbytis entellus). PhD Thesis, University of Jodhpur.
Rajpurohit, L. S. & Chhangani, A. K. (2003). Resident male change and infanticide in free-ranging unimale bisexual troop of Hanuman langur (Semnopithecus entellus) around Jodhpur (India). Proceedings of the 73rd Annual Session of the National Academy of Science, India, held at Ahmedabad in Oct, 2003 (pp. 72-73).
Rajpurohit, L. S., Chhangani, A. K,, Rajpurohit, R. S., & Mohnot, S. M. (2003). Observation of a sudden resident male replacement in a unimale bisexual troop of Hanuman langurs, Semnopithecus entellus, around Jodhpur (India). Folia Primatologica, 74, 85-87.
Roonwal, M. L., & Mohnot, S. M. (Eds.). (1977). Primates of South Asia: Ecology, sociobiology and behavior. Cambridge: Harvard University Press.
Ross, C. (1993). Take-over and infanticide in South Indian Hanuman langurs (Presbytis entellus). American Journal of Primatology, 30, 75-82.
Rudran, R. (1973). Adult male replacement in one-male troops of purple-faced langurs (Presbytis senex senex) and its effect on population structure. Folia Primatologica, 19, 166-192.
Sommer, V., & Rajpurohit, L. S. (1989), Male reproductive success in harem troops of Hanuman langurs (Presbytis entellus). International Journal of Primatology, 100, 293-317.
Sommer, V., & Mohnot, S. M. (1985). New observations on infanticide among Hanuman langurs (Presbytis entellus) near Jodhpur (Rajasthan, India). Behaviour, Ecology and Sociobiology, 16, 245-248.
Struhsaker, T. T., & Leland, L. (1985) Infanticide in a patrilineal society of red colobus monkeys. Zeitschrift f�r Tierpsychologie, 69, 89-132.
Sugiyama, Y. (1965a). Behavioral development and social structure in two troops of Hanuman langurs (Presbytis entellus). Primates, 6, 213-247.
Sugiyama, Y. (1965b). On the social change of Hanuman langurs (Presbytis entellus) in their natural conditions. Primates, 6, 381-418.
Vogel, C., & Loch, H. (1984). Reproductive parameters, adult-male replacements and infanticide among free-ranging langurs (Presbytis entellus) at Jodhpur (Rajasthan), India. In G. Hausfater & S. B. Hrdy (Eds.), Infanticide: Comparative and evolutionary perspectives (pp. 237-256). Hawthorne, NY: Aldine.
Wolfheim, J. H. (1983). Primates of the world. Seattle, WA: University of Washington Press.
-------------------------------------------
Dr. Goutam Sharma, Animal Behaviour Unit, Dept of Zoology, Jai Narain Vyas University, Jodhpur�342005 (Rajasthan), India [e-mail: [email protected]].
-------------------------------------------
* * *
Grants Available: Fyssen Foundation Postdoctoral Research Grants
Priority will be given to researchers who will develop their project in a different laboratory from the one at which they received their doctorate and from the laboratories where they are working at present.
The research grant, for one year without renewal, could range from 15,000 to 30,000 Euros. The financing of equipment above 7,500 Euros is excluded. One year after the award, the researchers will have to provide a detailed financial report about the use of the grant, and a short scientific report in French.
Application details and forms may be obtained from the Secr�tariat de la Fondation Fyssen, 194, rue de Rivoli, 7500l Paris, France [e-mail: [email protected]]; or see <www.fondation-fyssen.org>. Fourteen copies of the complete proposal should be sent by post to the Secr�tariat (address above), and must be received by October 29, 2010.
* * *
F. Barbara Orlans, PhD, passed away on June 18, 2010, in her home in Bethesda, Maryland. The Scientists Center for Animal Welfare (SCAW) expresses sympathy to her family and acknowledges the loss of a major contributor to the improvement of humane care and treatment of research animals. Dr. Orlans was the Founding President of SCAW in 1984, later becoming the Center�s first Executive Director, and spent many years establishing SCAW as a nonprofit and educational organization. For more information, see <www.scaw.com>.
Monkeys Use Tree Branches to Escape Researchers
In a development that offers new insights into the intelligence of monkeys, animals at a research institute in Japan have used the branches of trees to catapult themselves over an electric fence to escape. A group of 15 monkeys at Kyoto University�s Primate Research Institute in Aichi Prefecture escaped from their forest home, which is surrounded by a five-meter electric fence. The monkeys made their break for freedom by bending and releasing tree branches to fling themselves one by one slingshot-fashion over the high-voltage fence. The pen held about 65 monkeys.
The trees were only two meters high, and there was up to three meters of open ground between the fence and the nearest trees to prevent monkeys from jumping over the fence from a tree. �Their jumping power was greater than we thought,� said Hirohisa Hirai, deputy head of the Institute. �We were extremely surprised by the intelligence and the power they used in order to escape.�
Despite the intelligence demonstrated by their great escape, the primates then appeared unsure what to do with their freedom. They remained by the gates of the fenced area and were lured back by scientists with peanuts.
�We think that maybe there was some kind of dispute among the monkeys in the forest, and so this group decided to leave,� Hirai said. �Fortunately, they stayed by the fence after escaping as they probably wanted to stay near to the other monkeys.� Scientists have trimmed the trees further in order to prevent a repeat escape.
The Kyoto institution is one of the world�s leading primate research centers and has produced a series of internationally recognized studies exploring the social interaction, behavior, and evolution of primates. � by Danielle Demetriou, Daily Telegraph, July 8, 2010 � The Vancouver Sun
Wide-Eyed Primate Photographed for First Time
A �cute� primate so rare it was thought to be extinct has been caught on camera in the forests of Sri Lanka for the first time, scientists said Monday. The Horton Plains slender loris is a small, nocturnal animal which can grow up to 17 centimetres (six inches) long, with big, bulging eyes.
Endemic to Sri Lanka, it was first discovered in 1937 but had only been seen four times since then. Scientists last caught a glimpse of the primate in 2002, and believed the elusive animal had since died out. But field researchers, working with the Zoological Society of London (ZSL), managed to track down the mysterious creature in the forests of central Sri Lanka. In a world first, they were able to take pictures of an adult male slender loris sitting on a tree branch. The field team was able to capture one of the creatures and give it a physical examination, the first time that has ever been done, before releasing it back into the wild.
But experts warned that deforestation in Sri Lanka �- largely blamed on the drive to create tea plantations in the region � was now the biggest threat to the loris. Craig Turner, a conservation biologist at the ZSL, said their natural forest habitat had been divided up for farming and logging use, cutting off the �very cute� animals from their partners. �The forest has now been fragmented into a series of small islands,� Turner told BBC radio. �They can�t move to one another, they can�t mate, breed, so it has real implications for the future persistence of the species.� � London (AFP), July 19
Mexico Arrests Man with 18 Monkeys
Mexican authorities have arrested a man who was trying to smuggle 18 small monkeys into the country by carrying them in his clothing. Roberto Sol Cabrera, a Mexican citizen, was stopped at a random check at Mexico City�s international airport after arriving from Lima.
In a statement, police said Mr. Sol Cabrera had been behaving �nervously�.
Once he was searched, it was discovered that he had hidden 18 titi monkeys in a girdle around his waist. After his arrest, Mr. Sol Cabrera confessed that the animals had travelled in his luggage, and that he had put them under his clothing �to protect them from X-rays� as he was going through customs. The animals had been put into socks, police explained, and two of them were dead at the time of confiscation.
Many species of titi monkeys, of the South American genus Callicebus, are on the CITES endangered animal list. The Mexican government recently restricted imports of primates and, since Mr. Sol Cabrera did not have any permits, he will remain in custody while more investigations take place. In a video published by the Mexican Public Security agency, Mr. Sol Cabrera says he had paid $30 for each specimen in Peru.
According to estimates, monkeys like the ones confiscated in the airport could have been sold for between $775 and $1,550 in Mexico.
Adrian Reuter, local representative for TRAFFIC � the wildlife trade monitoring network � told the BBC that animal trafficking is a serious problem in Mexico. �The reasons are two: one, because Mexico is an important route for those who want to smuggle animals into the U.S.; and the other, because, as in other countries of Latin America, there is a deep-rooted tradition of having wild animals as pets,� he said. The Sonora market, in the Mexican capital, is known to sell parrots, monkeys and reptiles to private owners. Mr. Reuter recognizes that in the last few years, the Mexican government has improved efforts on fighting animal-trafficking criminal networks, rather than focusing on the citizens who want to have �a parrot for company,� he says. � July 19, by Julian Miglierini, BBC News, Mexico
New Director at Wisconsin Primate Center
Jon Levine, a professor of neurobiology and physiology at Northwestern University since 1984, takes the reins of the Wisconsin National Primate Research Center (WNPRC) this month. He assumes leadership of a center that is one of the most intellectually stimulating and scientifically important on campus and, at the same time, one of the most controversial due to its mission of research involving nonhuman primates.
While the center, which has a colony of about 1,400 primates representing three species, mostly rhesus macaques, is a lightning rod for controversy about the nature of its work, it is unarguably a dynamic, front-line research center with top-flight programs of research on such things as HIV vaccine development, regenerative medicine and reproduction, and aging and metabolic disease.
Levine is acutely aware of the need for open and honest communication with the public and the Madison community, in particular. While he has no illusions about his ability to sway the most adamant critics of nonhuman primate research, he argues that there are many untold truths, chief among them that center staff are committed, hard working people dedicated to helping solve some of the most pressing biomedical issues of our day. � U.W. Madison News, Sept 8, by Terry Devitt
National Primate Liberation Week
Michael A. Budkie, Executive Director of SAEN (Stop Animal Exploitation NOW!), has announced that October 16 to 24 will be National Primate Liberation Week, 2010.
He writes, �Over 70,000 primates (rhesus monkeys, baboons, squirrel monkeys, chimpanzees, etc.) are experimented on every year in the United States. Many thousands of primates are also imprisoned in labs for breeding and conditioning. Recent USDA statistics reveal that over 124,000 primates are held captive in U.S. laboratories. These thousands of animals are depending on us to fight for their freedom.
�Protests, vigils, news conferences, informational tabling, leafleting, etc. are all worthwhile events. The type of event that you choose is limited only by your own imagination. Utilize the informational resources on the SAEN Website, <www.saenonline.org>, to help in your planning.�
* * *
* * *
They are asking $2500 for an Anprolene AN74i Gas Sterilization Unit (Figure 2); $7500 for a custom wheeled stainless steel primate transport chamber (62� H x 32� L x 32� W), made by Lab Products, Seaford, Delaware (Figure 3); and $250 for a large animal scale, Vet-Tec 300 (Technidyne).
 |
 |
 |
|---|---|---|
| Figure 1: Social Interaction Units | Figure 2: Gas Sterilization Unit | Figure 3: Primate Transport Chamber |
Contact Mike Smith, 403 BSB, 173 Ashley Ave, Charleston, SC 29425 [843-792-5289; fax: 843-792-4423; e-mail: [email protected]] if you are interested.
* * *
Thursday, Sept 16, 6:30 p.m.: Prof. Katharine Milton, of the Dept. of Environmental Science, Policy and Management, University of California, Berkeley, will speak on �Unfortunately, not all obligate parasites are benign: Howler monkey�bot fly interactions in Panama�.
Thursday, Oct 21: Prof. Tony Goldberg, of the Dept. of Pathobiological Sciences, University of Wisconsin, Madison, will speak on �Evolutionary ecology of infectious disease in the non-human primates, people, and livestock of western Uganda�.
Thursday, Dec 9: Prof. Julie Wieczkowski, of the Dept. of Anthropology, Buffalo State College, will speak on �Feeding ecology of a seed predator, the Tana River mangabey of Kenya�.
If you would like to have dinner with any of the speakers, please contact Prof. Jessica Rothman <[email protected]>.
* * *
Visit the Guidance section on the Office of Laboratory Animal Welfare (OLAW) Website for updated Commentary on Lab Animal Protocol Review columns. �A Word from OLAW and USDA� responds to the topic �Revisiting Approved Protocols�, in the June 2010 issue: <grants.nih.gov/grants/olaw/references/39_6_0610.pdf>. To further explore this and other topics, browse the OLAW Topic Index: <grants.nih.gov/grants/olaw/olaw_topic_index.htm>. For additional information, visit OLAW: <grants.nih.gov/grants/olaw/olaw.htm>.
New Enrichment and Social Housing Resources
OLAW has a new online resource for information on nonhuman primate enrichment and social housing: <grants.nih.gov/grants/olaw/primate_enrichment-social_housing.htm>, to assist institutions in enhancing the care and well-being of nonhuman primates. On this site, you can find new frequently asked questions; a special online seminar; the OLAW report on site visits to chimpanzee facilities; a bibliographic guide developed by USDA, the National Agricultural Library, and the Animal Welfare Information Center; and more. For questions, suggestions, or comments, e-mail < [email protected]>.
Database for Sharing Aging Research Models
The National Institute on Aging (NIA) has developed a Website to facilitate sharing of research models, tissue or data: the Database for Sharing Aging Research Models (D-SARM) <dsarm.niapublications.org>. The database was developed with mouse and rat models in mind, but it is not limited to rodent models. Investigators engaged in research on the biology of aging using model organisms are encouraged to list their models in the database and include relevant descriptors of the model, such as �calorie restricted�, �rapamycin treated�, etc.
The search function on the Website is open to all investigators, but a password is required to join the network and submit information on the models in your laboratory. Investigators who list model organisms have their identity protected by a self-chosen user-name, so that initial contact is blind, allowing the individual with the model in question to determine how to make contact with potential requestors.
Questions may be directed to Nancy L. Nadon, PhD, Chief, Biological Resources Branch, Biology of Aging Program, National Institute on Aging, 7201 Wisconsin Ave., Rm 2C231, Bethesda, MD 20892 [301-402-7744; fax: 301-402-5997; e-mail: [email protected]]. � Released by NIA July 22
Rhesus Photographs
Viktor Reinhardt has posted 282 original photo scans of rhesus macaques at <www.flickr.com/photos/13720232@N04>. They are organized as:
Total E-Clips is a daily newsletter, published by e-mail by the Foundation for Biomedical Research, covering the latest news of interest to the animal research community. It regularly features news of medical breakthroughs, updates on animal extremism, and important announcements from inside the Beltway. The newsletter is delivered directly to your e-mail in-box each weekday.
To receive Total E-Clips daily, send an e-mail to < [email protected]>. Your address will be added to their mailing list, which is not shared with any other firm or organization.
* * *
* * *
One key request of researchers across the world is unrestricted access to research publications. Open access serves a worldwide audience larger than that of any subscription-based journal, thus increases the visibility and impact of published work. It also enhances indexing and retrieval power, and eliminates the need for permissions to reproduce and distribute content. JNBH is fully committed to the Open Access Initiative and will provide free access to all articles as soon as they are published.
* * *
Three-Year Residency Program � Buffalo, New York
The Division of Comparative Medicine and Laboratory Animal Facilities at the State University of New York at Buffalo is seeking candidates for a three-year American College of Laboratory Animal Medicine (ACLAM)-accredited Residency Program in Laboratory Animal Medicine.
The Resident will acquire skills and experience relevant to completing requirements for the ACLAM board examinations. Duties include anesthesia, surgery, medicine, research support, Institutional Animal Care and Use Committee duties, teaching, and resource management. In addition, the Resident will be expected to participate in his/her own research project suitable for publication, and attend a didactic training program in comparative medicine and lab animal science. Opportunities to train at the local zoo and aquarium and nearby universities are available. One month is spent at the University of Texas M. D. Anderson Cancer Center to gain practical experience with nonhuman primates.
Financial support is provided to attend a conference each year. Benefits include health care and dental/eye care.
Successful candidates must be a graduate of an accredited school, have a DVM degree, and be eligible to obtain a New York state veterinary license. All applicants should submit a resume, letter of intention and goals, and contacts for three references. Academic transcripts will be required prior to acceptance into the program.
Further information is available at <www.research.buffalo.edu/laf/resources/vet_residency.cfm> or by contacting Dr. Lisa Martin [716-829-3877; e-mail:[email protected]].
* * *
�Why yes, we use human primates in our research. They are the best model,
because they are so closely related to us.�
* * *
Recent Books and Articles
(Addresses are those of first authors unless otherwise indicated)
Books
* Almost Chimpanzee: Searching for What Makes Us Human, in Rainforests, Labs, Sanctuaries, and Zoos. J. Cohen. New York: Times Books, 2010. 384 pp. [Price: $27.50]
* Animal Models for Retinal Diseases. I.-H. Pang & A. F. Clark (Eds.). Humana Press, distributed by Springer, 2010. [Price: $119]
* The Evolution of Language. W. T. Fitch. West Nyack, NY: Cambridge University Press, 2010. [Price: paper, $55; hardcover, $120]
* Indonesian Primates. S. Gursky-Doyen & J. Supriatna (Eds.). Developments in Primatology: Progress and Prospects. R. H. Tuttle (Series Ed.). New York: Springer, 2010. [Price: $169]
* The Last Stand of the Gorilla: Environmental Crime and Conflict in the Congo Basin. C. Nellemann, I. Redmond, & J. Refisch (Eds.). United Nations Environment Programme, GRID-Arendal, 2010. 88 pp. <www.grida.no/_res/site/file/publications/gorilla/GorillaStand_screen.pdf>.
* Multisensory Object Perception in the Primate Brain. M. J. Naumer & J. Kaiser (Eds.). New York: Springer, 2010. 350 pp. [Price: $159]
. . .
Contents: General introduction, by M. J. Naumer & J. Kaiser. Part I. Mechanisms. Corticocortical connectivity subserving different forms of multisensory convergence, by M. A. Meredith & H. R. Clemo; Computational modeling of multisensory object perception, by C. Rothkopf, T. Weisswange, & J. Triesch; Methodological considerations: Electrophysiology of multisensory interactions in humans, by M.-H. Giard & J. Besle; Cortical oscillations and multisensory interactions in humans, by J. Kaiser & M. J. Naumer; and Multisensory functional magnetic resonance imaging, by M. J. Naumer, J. J. F. van den Bosch, A. Polony, & J. Kaiser. Part II. Audio-visual Integration. Audiovisual temporal integration for complex speech, object-action, animal call, and musical stimuli, by A. Vatakis & C. Spence; Imaging cross-modal influences in auditory cortex, by C. Kayser, C. I. Petkov, & N. K. Logothetis; The default mode of primate vocal communication and its neural correlates, by A. A. Ghazanfar; Audio-visual perception of everyday natural objects � Hemodynamic studies in humans, by J. W. Lewis; and Single-trial multisensory learning and memory retrieval, by M. M. Murray & H. F. Sperdin. Part III. Visuo-tactile integration. Multisensory texture perception, by R. L. Klatzky & S. J. Lederman; Dorsal and ventral cortical pathways for visuo-haptic shape integration revealed using fMRI, by T. W. James & S. Kim; Visuo-haptic perception of objects and scenes, by F. N. Newell; and Haptic face processing and its relation to vision, by S. J. Lederman, R. L. Klatzky, & R. Kitada. Part IV. Plasticity. The ontogeny of human multisensory object perception: A constructivist account, by D. J. Lewkowicz; Neural development and plasticity of multisensory representations, by M. T. Wallace, J. Krueger, & D. W. Royal; and Large-scale brain plasticity following blindness and the use of sensory substitution devices, by A. Bubic, E. Striem-Amit, & A. Amedi.
* The Red Colobus Monkeys: Variation in Demography, Behavior, and Ecology of Endangered Species. T. T. Struhsaker. New York: Oxford University Press, 2010. 349 pp. [Price: $99]
* The UFAW Handbook on the Care and Management of Laboratory and Other Research Animals, 8th ed. R. Hubrecht & J. Kirkwood (Eds.). Hoboken, NJ: Wiley-Blackwell, 2010. [Price: $399.99]
* Primates of West Africa: Pocket Identification Guide. J. F. Oates & S. D. Nash (Illus.). Conservation International, 2010. 20 pp. [Price: $7.95], <www.primate-sg.org/pocketPWAFG.htm>.
* A Field Guide to the Mammals of Central America and Southeast Mexico (2d ed.). F. A. Reid. New York: Oxford University Press, 2009. 384 pp. [Price: paper, $45; hard-cover, $149]
* American Journal of Primatology, 2010, 72[6], <www3.interscience.wiley.com/journal/123371344/issue>.
. . .Contents: Baboon vaginal microbiota: An overlooked aspect of primate physiology, by A. J. Rivera, R. M. Stumpf, B. Wilson, S. Leigh, & A. A. Salyers; Impact of yellow fever outbreaks on two howler monkey species (Alouatta guariba clamitans and A. caraya) in Misiones, Argentina, by I. Holzmann, I. Agostini, J. I. Areta, H. Ferreyra, P. Beldomenico, & M. S. Di Bitetti; Dental wear, wear rate, and dental disease in the African apes, by A. A. Elgart; Using behavior to determine immature life-stages in captive western gorillas, by J. E. Hutchinson & A. W. Fletcher; Prey capture efficiency in brown capuchin monkeys (Cebus apella) is influenced by sex and corpus callosum morphology, by K. Hellner-Burris, C. A. Sobieski, V. R. Gilbert, & K. A. Phillips; Cannibalism in wild bonobos (Pan paniscus) at Lui Kotale, by A. Fowler & G. Hohmann; Feeding ecology of a group of buffy-headed marmosets (Callithrix flaviceps): Fungi as a preferred resource, by R. R. Hil�rio & S. F. Ferrari; Lactational programming? Mother�s milk energy predicts infant behavior and temperament in rhesus macaques (Macaca mulatta), by K. Hinde & J. P. Capitanio; Validation of multi-detector computed tomography as a non-invasive method for measuring ovarian volume in macaques (Macaca fascicularis), by J. C. Jones, S. E. Appt, S. R. Werre, J. C. Tan, & J. R. Kaplan; Gastrointestinal parasites of howler monkeys (Alouatta palliata) inhabiting the fragmented landscape of the Santa Marta mountain range, Veracruz, Mexico, by C. Valdespino, G. Rico-Hern�ndez, & S. Mandujano; and Neighbor effects in marmosets: Social contagion of agonism and affiliation in captive Callithrix jacchus, by C. F. I. Watson & C. A. Caldwell.
* American Journal of Primatology, 2010, 72[7], <www3.interscience.wiley.com/journal/123456845/issue>.
. . .Contents: Effects of reproductive condition and dominance rank on cortisol responsiveness to stress in free-ranging female rhesus macaques, by C. L. Hoffman, J. E. Ayala, A. Mas-Rivera, & D. Maestripieri; Fecal bacterial diversity of human-habituated wild chimpanzees (Pan troglodytes schweinfurthii) at Mahale Mountains National Park, Western Tanzania, by B. A. Szekely, J. Singh, T. L. Marsh, C. Hagedorn, S. R. Werre, & T. Kaur; Nest groups of wild bonobos at Wamba: Selection of vegetation and tree species and relationships between nest group size and party size, by M. N. Mulavwa, K. Yangozene, M. Yamba-Yamba, B. Motema-Salo, N. N. Mwanza, & T. Furuichi; Genetic characterization of specific pathogen-free rhesus macaque (Macaca mulatta) populations at the California National Primate Research Center (CNPRC), by S. Kanthaswamy, A. Kou, J. Satkoski, M. C. T. Penedo, T. Ward, J. Ng, L. Gill, N. W. Lerche, B. J.-A. Erickson, & D. G. Smith; Sources of variation in fecal cortisol levels in howler monkeys in Belize, by A. M. Behie, M. S. M. Pavelka, & C. A. Chapman; Vegetation correlates of gibbon density in the peat-swamp forest of the Sabangau catchment, Central Kalimantan, Indonesia, by M. Hamard, S. M. Cheyne, & V. Nijman; Selection of sleeping trees in pileated gibbons (Hylobates pileatus), by R. Phoonjampa, A. Koenig, C. Borries, G. A. Gale, & T. Savini; Effects of early rearing conditions on problem-solving skill in captive male chimpanzees (Pan troglodytes), by N. Morimura & Y. Mori; Changes in ranging and agonistic behavior of vervet monkeys (Cercopithecus aethiops) after predator-induced group fusion, by K. Enstam Jaffe & L. A. Isbell; and Status of diurnal primate populations at the former settlement of a displaced village in Cameroon, by L. Baya & I. Storch.
* Animal Behaviour, 2010, 79[1], <www.sciencedirect.com/science/journal/00033472>.
. . .Contents include: A domain-specific opposite-sex bias in human preferences for manipulated voice pitch, by B. C. Jones, D. R. Feinberg, L. M. DeBruine, A. C. Little, & J. Vukovic; Multiple resource values and fighting ability measures influence intergroup conflict in guerezas (Colobus guereza), by T. R. Harris; Indirect social influence in the maintenance of the stone-handling tradition in Japanese macaques, Macaca fuscata, by J.-B. Leca, N. Gunst, & M. A. Huffman; Loud calls in male crested macaques, Macaca nigra: A signal of dominance in a tolerant species, by C. Neumann, G. Assahad, K. Hammerschmidt, D. Perwitasari-Farajallah, & A. Engelhardt; and Face scanning in chimpanzees and humans: Continuity and discontinuity, by F. Kano & M. Tomonaga.
* Animal Behaviour, 2010, 79[2], <www.sciencedirect.com/science/journal/00033472>.
. . .Contents include: Bearded capuchin monkeys� and a human�s efficiency at cracking palm nuts with stone tools: Field experiments, by D. Fragaszy, T. Pickering, Q. Liu, P. Izar, E. Ottoni, & E. Visalberghi; Male aggression and sexual coercion in wild West African chimpanzees, Pan troglodytes verus, by R. M. Stumpf & C. Boesch; Inferring networks from multivariate symbolic time series to unravel behavioural interactions among animals, by D. M. Walker, C. Carmeli, F. J. P�rez-Barber�a, M. Small, & E. P�rez-Fern�ndez; Resting time as an ecological constraint on primate biogeography, by A. H. Korstjens, J. Lehmann, & R. I. M. Dunbar; A method for exploring the structure of behavioural syndromes to allow formal comparison within and between data sets, by N. J. Dingemanse, N. Dochtermann, & J. Wright; and Evidence of sexually selected infanticide in an endangered brown bear population, by A. Fern�ndez-Gil, J. E. Swenson, C. Granda, T. P�rez, A. Dom�nguez, A. Ordiz, J. Naves, & M. Delibes.
* Animal Behaviour, 2010, 79[3], <www.sciencedirect.com/science/journal/00033472>.
. . .Contents include: The influence of kin relationship and reciprocal context on chimpanzees� other-regarding preferences, by S. Yamamoto & M. Tanaka; Choosing an appropriate index to construct dominance hierarchies in animal societies: A comparison of three indices, by A. Bang, S. Deshpande, A. Sumana, & R. Gadagkar; and Wilcoxon signed-ranks test: Symmetry should be confirmed before the test, by E. Kasuya.
* Animal Behaviour, 2010, 79[4], <www.sciencedirect.com/science/journal/00033472>.
. . .Contents include: Leadership and social information use in human crowds, by J. J. Faria, J. R. G. Dyer, C. R. Tosh, & J. Krause; and Is genetic diversity associated with mating success in humans? By H. C. Lie, G. Rhodes, & L. W. Simmons.
* Animal Behaviour, 2010, 79[5], <www.sciencedirect.com/science/journal/00033472>.
. . .Contents include: Hedging their bets? Male and female chacma baboons form friendships based on likelihood of paternity, by L. R. Moscovice, A. Di Fiore, C. Crockford, D. M. Kitchen, R. Wittig, R. M. Seyfarth, & D. L. Cheney; Grooming for infant handling in tufted capuchin monkeys: A reappraisal of the primate infant market, by B. Tiddi, F. Aureli, & G. Schino; and Recognition of faces of known individuals in two lemur species (Eulemur fulvus and E. macaco), by L. Marechal, E. Genty, & J. J. Roeder.
* Animal Behaviour, 2010, 79[6], <www.sciencedirect.com/science/journal/00033472>.
. . .Contents include: Infants as costly social tools in male Barbary macaque networks, by S. Henkel, M. Heistermann, & J. Fischer; Resting and nesting in a small mammal: Sleeping sites as a limiting resource for female grey mouse lemurs, by H. Lutermann, L. Verburgt, & A. Rendigs; Mechanisms underlying responses to inequitable outcomes in chimpanzees, Pan troglodytes, by S. F. Brosnan, C. Talbot, M. Ahlgren, S. P. Lambeth, & S. J. Schapiro; and One cultural parent makes no culture, by M. Enquist, P. Strimling, K. Erikkson, K. Laland, & J. Sjostrand.
* Animal Cognition, 2010, 13[3], <www.springerlink.com/content/101775>.
. . .Contents include: Spotting animals in natural scenes: Efficiency of humans and monkeys at very low contrasts, by M. J.-M. Mac�, A. Delorme, G. Richard, & M. Fabre-Thorpe; Bonobos, chimpanzees, gorillas, and orang utans use feature and spatial cues in two spatial memory tasks, by P. Kanngiesser & J. Call; The apes� edge: Positional learning in chimpanzees and humans, by A. D. Endress, S. Carden, E. Versace, & M. D. Hauser; and Perception of biological motion in common marmosets (Callithrix jacchus): By females only, by J. Brown, G. Kaplan, L. J. Rogers, & G. Vallortigara.
* Animal Cognition, 2010, 13[4], <www.springerlink.com/content/101775>.
. . .Contents include: Triadic and collaborative play by gorillas in social games with objects, by J. E. Tanner & R. W. Byrne; Characteristics of implicit chaining in cotton-top tamarins (Saguinus oedipus), by C. Locurto, M. Gagne, & L. Nutile; A modified mark test for own-body recognition in pig-tailed macaques (Macaca nemestrina), by S. Macellini, P. F. Ferrari, L. Bonini, L. Fogassi, & A. Paukner; Chimpanzees (Pan troglodytes) accurately compare poured liquid quantities, by M. J. Beran; Erratum to: Chimpanzees (Pan troglodytes) accurately compare poured liquid quantities, by M. J. Beran; and Donor payoffs and other-regarding preferences in cotton-top tamarins (Saguinus oedipus), by J. R. Stevens.
* Experimental Animals, 2010, 59[2], <www.jstage.jst.go.jp/browse/expanim/59/2/_contents>.
. . .Contents include: Comparative proteomic analyses of macular and peripheral retina of cynomolgus monkeys (Macaca fascicularis), by H. Okamoto, S. Umeda, T. Nozawa, M. T. Suzuki, Y. Yoshikawa, E. T. Matsuura, & T. Iwata; Female reproduction characteristics in a large-scale breeding colony of cynomolgus monkeys (Macaca fascicularis), by T. Yoshida, K. Hanari, K. Fujimoto, & T. Sankai; and Fetal sex determination of macaque monkeys by a nested PCR using maternal plasma, by F. Mitsunaga, M. Ueiwa, Y. Kamanaka, M. Morimoto, & S. Nakamura.
* Folia Primatologica, 2010, 81[1], <www.karger.com/fpr>.
. . .Contents: Altitudinal ranging of black-crested gibbons at Mt. Wuliang, Yunnan: Effects of food distribution, temperature and human disturbance, by P.-F. Fan & X.-L. Jiang; Cross-amplified polymorphic microsatellites for Campbell�s monkey, by E. J. Petit, K. Ouattara, K. Zuberb�hler, D. Vallet, & A. Lemasson; A capuchin monkey (Cebus apella) uses video to find food, by P. Pot� & M. Saporiti; Four breeding females in a free-ranging group of buffy-headed marmosets (Callithrix flaviceps), by R. R. Hil�rio & S. F. Ferrari; and Postdispersal removal and germination of seed dispersed by Cercopithecus nictitans in a West African montane forest, by H. M. Chapman, S. L. Goldson, & J. Beck.
* Folia Primatologica, 2010, 81[2], <www.karger.com/fpr>.
. . .Contents: Nasal anatomy of Paradolichopithecusgansuensis (early Pleistocene, Longdan, China) with comments on phyletic relationships among the species of this genus, by T. D. Nishimura, Y. Zhang, & M. Takai; Insect-foraging in captive owl monkeys (Aotus nancymaae), by C. K. Wolovich, J. Rivera, & S. Evans; Functional context of scent-marking in Callithrix penicillata, by D. G. R. Oliveira & R. H. Macedo; Predation and predation attempts on red titi monkeys (Callicebus discolor) and equatorial Sakis (Pithecia aequatorialis) in Amazonian Ecuador, by A. G. de Luna, R. Sanmiguel, A. Di Fiore, & E. Fernandez-Duque; and Postrelease success of two rehabilitated vervet monkey (Chlorocebus aethiops) troops in KwaZulu-Natal, South Africa, by K. Wimberger, C. T. Downs, & M. R. Perrin.
* International Journal of Primatology, 2010, 31[3], <www.springerlink.com/content/104389>.
. . .Contents: Diet, activity patterns, and ranging ecology of the Bale monkey (Chlorocebus djamdjamensis) in Odobullu Forest, Ethiopia, by A. Mekonnen, A. Bekele, P. J. Fashing, G. Hemson, & A. Atickem; Ranging patterns of two syntopic howler monkey species (Alouatta guariba and A. caraya) in northeastern Argentina, by I. Agostini, I. Holzmann, & M. S. Di Bitetti; High population density of Pan troglodytes verus in the Haut Niger National Park, Republic of Guinea: Implications for local and regional conservation, by M.-C. Fleury-Brugiere & D. Brugiere; Fruit preferences of Ateles belzebuth in Tinigua Park, northwestern Amazonia, by P. R. Stevenson & A. Link; Infanticide in Cercopithecus mitis stuhlmanni in the Kakamega Forest, Kenya: Variation in the occurrence of an adaptive behavior, by M. Cords & J. L. Fuller; Life history of Cercopithecus mitis stuhlmanni in the Kakamega Forest, Kenya, by M. Cords & S. Chowdhury; Wild capuchins show male-biased feeding tool use, by A. C. de A. Moura & P. C. Lee; Use of mineral licks by white-bellied spider monkeys (Ateles belzebuth) and red howler monkeys (Alouatta seniculus) in eastern Ecuador, by J. G. Blake, J. Guerra, D. Mosquera, R. Torres, B. A. Loiselle, & D. Romo; and Line transect sampling of primates: Can animal-to-observer distance methods work? By S. T. Buckland, A. J. Plumptre, L. Thomas, & E. A. Rexstad.
* International Journal of Primatology, 2010, 31[4], <www.springerlink.com/content/104389>.
. . .Contents: Reproductive and life history parameters of wild female Macaca assamensis, by I. F�rtbauer, O. Sch�lke, M. Heistermann, & J. Ostner; Are there ubiquitous parasite-driven major histocompatibility complex selection mechanisms in gray mouse lemurs? by N. Schwensow, M. Eberle, & S. Sommer; Sex differences in the song of Indri indri, by C. Giacoma, V. Sorrentino, C. Rabarivola, & M. Gamba; Seasonal variation in seed dispersal by tamarins alters seed rain in a secondary rain forest, by L. Culot, F. J. J. Mu�oz Lazo, M.-C. Huynen, P. Poncin, & E. W. Heymann; Activity patterns of Brachyteles arachnoides in the largest remaining fragment of Brazilian Atlantic forest, by M. G. Talebi & P. C. Lee; Orangutan energetics and the influence of fruit availability in the nonmasting peat-swamp forest of Sabangau, Indonesian Borneo, by M. E. Harrison, H. C. Morrogh-Bernard, & D. J. Chivers; Effects of seasonal folivory and frugivory on ranging patterns in Rhinopithecus roxellana, by Y. Li, Z. Jiang, C. Li, & C. C. Grueter; Home-range use by a large horde of wild Mandrillus sphinx, by E. C. White, J.-T. Dikangadissi, E. Dimoto, W. B. Karesh, M. D. Kock, N. O. Abiaga, R. Starkey, T. Ukizintambara, L. J. T. White, & K. A. Abernethy; Adaptation to forest life during the reintroduction process of immature Pongo abelii, by B. Riedler, E. Millesi, & P. H. Pratje; and Decrease in alarm call response among tufted capuchins in competitive feeding contexts: Possible evidence for counterdeception, by B. C. Wheeler.
* IPPL News, May, 2010, 37[1], <www.ippl.org/news.php>. (Intl Primate Protection League, P.O. Box 766, Summerville, SC 29484 [e-mail: [email protected]]).
. . .Contents include: Socializing captive-born Taiwanese macaques, by J. Spowart; and items from the IPPL 2010 Members� Meeting.
* Journal of the American Association for Laboratory Animal Science, 2010, 49[4], <tinyurl.com/2ukbxj8>.
. . .Contents include: Survey of prevalence of overweight body condition in laboratory-housed cynomolgus macaques (Macaca fascicularis), by S. A. Bauer, K. E. Leslie, D. L. Pearl, J. Fournier, & P. V. Turner; and Modified skin-window technique in cynomolgus macaques (Macaca fasicularis) for assessing neutrophil extravasation, by S. Maguire, E. Dobrzynski, & R. K. Alpaugh.
* Journal of Medical Primatology, 2010, 39[3], <www3.interscience.wiley.com/journal/123420592/issue>.
. . .Contents: Separate and combined effects of caffeine and dbcAMP on olive baboon (Papio anubis) sperm, by A. Nyachieo, C. Spiessens, D. C. Chai, N. M. Kiulia, J. M. Mwenda, & T. M. D�Hooghe; The normal and abnormal owl monkey (Aotus sp.) heart: Looking at cardiomyopathy changes with echocardiography and electrocardiography, by R. S. Rajendra, A. G. Brady, V. L. Parks, C. V. Massey, S. V. Gibson, & C. R. Abee; Experience with a naphthylmedetomidine � ketamine � hyaluronidase combination in inducing immobilization in anthropoid apes, by L. Hess, M. Votava, J. Schreiberov�, J. M�lek, & M. Hor�ček; Epizootic fatal amebiasis in an outdoor group of Old World monkeys, by R. Ulrich, M. B�er, V. Herder, I. Spitzbarth, M. Hewicker-Trautwein, W. Baumg�rtner, & P. Wohlsein; Congenital digital aplasia in a free-ranging group of silvery marmosets, Mico argentatus, by S. F. Ferrari, P. E. G. Coutinho, & H. K. M. Corr�a; Ovarian pathology in rhesus macaques: A 12-year retrospective, by A. K. Marr-Belvin, C. C. Bailey, H. L. Knight, S. A. Klumpp, S. V. Westmoreland, & A. D. Miller; Hematological, biochemical, respiratory, cardiovascular and electroneurophysiological parameters in African green monkeys (Cercopithecus aethiops sabaeus). Its use in non-clinical toxicological studies, by A. Casac�, I. Beausoleil, B. Gonzalez, P. Luaces, A. Le�n, M. E. Arteaga, P. Prado, V. Rodr�guez, Ayl�n P�rez, G. Guevara, A. M. Bada, N. Led�n, D. Fuentes, C. Gonz�lez, O. Hernandez, R. Orphee, D. Blanco, M. Garc�a-Osuma, & A. Ballester-Labrada; and Indication of higher salivary α-amylase expression in hamadryas baboons and geladas compared to chimpanzees and humans, by M. Mau, K.-H. S�dekum, A. Johann, A. Sliwa, & T. M. Kaiser.
* Primarily Primates Newsletter, Summer 2010, <primarilyprimates.org/newsletter>.
* Primate Research, 2010, 26[1], <www.jstage.jst.go.jp/browse/psj/26/1/_contents>.
. . .Contents: Identification of the tooth class of molars in Japanese macaques (Macaca fuscata) by using a geometric morphometric method, by T. Ito, S. Ogiono, Y. Nishioka, & M, Takai; A mathematical model for tamarin�s tool-use behavior, by T. Mariyama, H. Itoh, & K. Nakamura; Aggressive response of Japanese macaques toward a Japanese giant flying squirrel, by K. Onishi, K. Yamada, & M. Nakamichi; Histological characteristics of spermatogenesis in testes of the orangutan (Pongo pygmaeus), by M. Nakano, H. Fujii-Hanamoto, T. Enomoto, K. Matsubayashi, & H. Kusunoki; and Pedological analysis of geophagic behaviour in captive Borneo orangutan (Pongo pygmaeus), by S. Yamazaki, S. Takeda, E. Torii, S. Suzuki, M. Shimizu, & H. Kurotori.
* Primates, 2010, 51[1], <www.springerlink.com/content/nj041v115442>.
. . .Contents: Heritability of digit ratio (2D:4D) in rhesus macaques (Macaca mulatta), by E. Nelson & M. Voracek; Preference for consonant music over dissonant music by an infant chimpanzee, by T. Sugimoto, H. Kobayashi, N. Nobuyoshi, Y. Kiriyama, H. Takeshita, T. Nakamura, & K. Hashiya; The first case of dental flossing by a Japanese macaque (Macaca fuscata): Implications for the determinants of behavioral innovation and the constraints on social transmission, by J.-B. Leca, N. Gunst, & M. A. Huffman; Locomotor evolution of Mesopithecus (Primates: Colobinae) from Greece: Evidence from selected astragalar characters, by D. Youlatos & G. D. Koufos; Of how much concern are the �least concern� species? Distribution and conservation status of bonnet macaques, rhesus macaques and Hanuman langurs in Karnataka, India, by H. N. Kumara, S. Kumar, & M. Singh; Behavioral modifications in northern bearded saki monkeys (Chiropotes satanas chiropotes) in forest fragments of central Amazonia, by S. A. Boyle & A. T. Smith; Genetic architecture of two red ruffed lemur (Varecia rubra) populations of Masoala National Park, by V. R. Razakamaharavo, S. M. McGuire, N. Vasey, E. E. Louis, & R. A. Brenneman; Visual preference in a human-reared agile gibbon (Hylobates agilis), by M. Tanaka & M. Uchikoshi; Entamoeba histolytica and E. dispar infections in captive macaques (Macaca fascicularis) in the Philippines, by W. L. Rivera, J. A. D. L. Yason & D. E. V. Adao; Stochastic variation in sex ratios in infant mortality rates due to small samples in provisioned Japanese macaque (Macaca fuscata) populations, by H. Kurita; Third-party grooming in a captive chimpanzee group, by Y. I. Russell; Infanticide following male takeover event in Verreaux�s sifaka (Propithecus verreauxi verreauxi), by B. L. Littlefield; Coprophagy in wild bonobos (Pan paniscus) at Wamba in the Democratic Republic of the Congo: A possibly adaptive strategy? by T. Sakamaki; and Coalitions and male�male behavior in Alouatta palliata, by P. A. D. Dias, A. Rangel-Negrin, J. J. Ve�, & D. Canales-Espinosa.
* Primates, 2010, 51[2], <www.springerlink.com/content/l781086v1011>.
. . .Contents: Coprophagy-related interspecific nocturnal interactions between Japanese macaques (Macaca fuscata yakui) and sika deer (Cervus nippon yakushimae), by M. Nishikawa & K. Mochida; An evaluation of the geographic method for recognizing innovations in nature, using zoo orangutans, by S. R. Lehner, J. M. Burkart, & C. P. van Schaik; Modelling ranging behaviour of female orang-utans: A case study in Tuanan, Central Kalimantan, Indonesia, by F. M. Wartmann, R. S. Purves, & C. P. van Schaik; Community ecology of the Middle Miocene primates of La Venta, Colombia: The relationship between ecological diversity, divergence time, and phylogenetic richness, by B. C. Wheeler; What defines successful integration into a social group for hand-reared chimpanzee infants? By M. J. Bashaw, R. L. Gullott, & E. C. Gill; Seasonal changes in food resource distribution and feeding sites selected by Japanese macaques on Koshima Islet, Japan, by M. Go; Effects of vegetation type on habitat use by crop-raiding Japanese macaques during a food-scarce season, by A. Yamada & Y. Muroyama; High prevalence of antibodies against hepatitis A virus among captive nonhuman primates, by P. Sa-nguanmoo, N. Thawornsuk, P. Rianthavorn, A. Sommanustweechai, P. Ratanakorn, & Y. Poovorawan; Old grandmothers provide essential care to their young granddaughters in a free-ranging group of Japanese monkeys (Macaca fuscata), by M. Nakamichi, K. Onishi, & K. Yamada; Do chimpanzees (Pan troglodytes) use cleavers and anvils to fracture Treculia africana fruits? Preliminary data on a new form of percussive technology, by K. Koops, W. C. McGrew, & T. Matsuzawa; Nonaggressive interventions by third parties in conflicts among captive Bornean orangutans (Pongo pygmaeus), by T. Tajima & H. Kurotori; and Case report of helminths and lung mite infection in the red-tailed monkey, Cercopithecus ascanius schmidti, in Mahale Mountains National Park, Tanzania, by T. Kooriyama, A. Inaba, T. Nishida, & T. Iwaki.
* Primates, 2010, 51[3], <www.springerlink.com/content/q0268212g481>.
. . .Contents: Behavior of geladas and other endemic wildlife during a desert locust outbreak at Guassa, Ethiopia: Ecological and conservation implications, by P. J. Fashing, N. Nguyen, & N. J. Fashing; Do bonobos say NO by shaking their head? by C. Schneider, J. Call, & K. Liebal; Reconciling conflicts in a one-male society: The case of geladas (Theropithecus gelada), by A. Leone & E. Palagi; Diet and feeding patterns in the kipunji (Rungwecebus kipunji) in Tanzania�s Southern Highlands: A first analysis, by T. R. B. Davenport, D. W. De Luca, C. E. Bracebridge, S. J. Machaga, N. E. Mpunga, O. Kibure, & Y. S. Abeid; Preliminary study of mother�offspring conflict in black and gold howler monkeys (Alouatta caraya), by R. Pav�, M. M. Kowalewski, S. M. Peker, & G. E. Zunino; Hybridization between Alouatta caraya and Alouatta guariba clamitans in captivity, by A. de Souza Jesus, H. E. Schunemann, J. M�ller, M. Ansolch da Silva, & J. C. Bicca-Marques; Parasitic infections of three Mexican howler monkey groups (Alouatta palliata mexicana) living in forest fragments in Mexico, by J. Crist�bal-Azkarate, B. Hervier, S. Vegas-Carrillo, D. Osorio-Sarabia, E. Rodr�guez-Luna, & J. J. Ve�; Secondary transfer of adult mantled howlers (Alouatta palliata) on Hacienda La Pacifica, Costa Rica: 1975�2009, by M. R. Clarke & K. E. Glander; Handedness in captive gorillas (Gorilla gorilla), by R. M. Harrison & P. Nystrom; and Prenatal ontogeny of subspecific variation in the craniofacial morphology of the Japanese macaque (Macaca fuscata), by W. Yano, N. Egi, T. Takano, & N. Ogihara.
* Veterinary Anaesthesia and Analgesia, 2010, 37[5], <onlinelibrary.wiley.com/doi/10.1111/j.1467-2995.2010.00561a.x/full>.
. . .Contents include Abstracts presented at the 34th American College of Veterinary Anesthesiologists Annual Meeting, 9th�12th September 2009, Chicago, IL.
* The 57th Annual Meeting of the Japanese Association for Laboratory Animal Science. Experimental Animals, 2010, 59[3], 271-394, <www.jstage.jst.go.jp/browse/expanim/59/3/_contents>.
* An assessment of trade in gibbons and orang-utans in Sumatra, Indonesia: A TRAFFIC Southeast Asia report. V. Nijman. TRAFFIC Southeast Asia, 2009. 57 pp. <www.worldwildlife.org/who/media/press/2009/WWFBinaryitem12135.pdf>.
* Environmental Enrichment for Nonhuman Primates Resource Guide. AWIC Resource Series No. 32, 2006 (Updated March 2010), <www.nal.usda.gov/awic/pubs/Primates2009/intro.shtml>.
. . .Contains the USDA Final Report on Environmental Enhancement to Promote the Psychological Well-Being of Nonhuman Primates and the USDA Draft Policy on Environmental Enrichment for Nonhuman Primates.
* Special Issue on Conservation Education. F. Dolins, A. Jolly, & A. Savage (Eds.). American Journal of Primatology, 2010, 72[5], <www3.interscience.wiley.com/journal/123329834/issue>.
. . .Contents: Introduction to this special issue of the American Journal of Primatology on conservation education, by P. A. Garber; Developing an effective community conservation program for cotton-top tamarins (Saguinus oedipus) in Colombia, by A. Savage, R. Guillen, I. Lamilla, & L. Soto; Conservation education in Madagascar: Three case studies in the biologically diverse island-continent, by F. L. Dolins, A. Jolly, H. Rasamimanana, J. Ratsimbazafy, A. T. C. Feistner, & F. Ravoavy; Evaluating for long-term impact of an environmental education program at the Kalinzu Forest Reserve, Uganda, by C. W. Kuhar, T. L. Bettinger, K. Lehnhardt, O. Tracy, & D. Cox; Effective primate conservation education: Gaps and opportunities, by S. K. Jacobson; Conservation education and primates: Twenty-first century challenges and opportunities, by H. M. Sherrow; Increasing the impact of conservation projects, by L. A. Dietz, M. Brown, & V. Swaminathan; Summary of recommendations for primate conservation education programs, by J. Wallis, & E. V. Lonsdorf; Discovering the unexpected: Lessons learned from evaluating conservation education programs in Africa, by T. L. Bettinger, C. W. Kuhar, K. Lehnhardt, D. Cox, & D. Cress; Primate conservation: Integrating communities through environmental education programs, by S. M. Padua; Education for the conservation of great apes and other wildlife in northern Congo � The importance of nature clubs, by T. Breuer & F. B. Mavinga; and Use of film for community conservation education in primate habitat countries, by J. H. Wright.
* Special AIDS Issue, Journal of Medical Primatology, 2010, 39[4], <onlinelibrary.wiley.com/doi/10.1111/jmp.2010.39.issue-4/issuetoc>.
. . .Contents: Introduction, by R. S. Veazey, A. Kaur, & D. T. Evans; In memory of Jonathan Sawyer Allan, MSc, DVM, by V. Hirsch; Primate-specific regulation of natural killer cells, by P. Parham, L. Abi-Rached, L. Matevosyan, A. K. Moesta, P. J. Norman, A. M. Older Aguilar, & L. A. Guethlein; Generation of a dual RT Env SHIV that is infectious in rhesus macaques, by J. M. Smith, A. Dauner, B. Li, P. Srinivasan, J. Mitchell, M. Hendry, D. Ellenberger, S. Butera, & R. A. Otten; Heterogeneity in phenotype and function of CD8+ and CD4/CD8 double-negative Natural Killer T cell subsets in sooty mangabeys, by N. Rout, J. G. Else, S. Yue, M. Connole, M. A. Exley, & A. Kaur; In acute pathogenic SIV infection plasmacytoid dendritic cells are depleted from blood and lymph nodes despite mobilization, by S. M. Barratt-Boyes, V. Wijewardana, & K. N. Brown; Microbial translocation in simian immunodeficiency virus (SIV)-infected rhesus monkeys (Macaca mulatta), by C. Leinert, C. Stahl-Hennig, A. Ecker, T. Schneider, D. Fuchs, U. Sauermann, & S. Sopper; Detecting signatures of inter-regional and inter-specific hybridization among the Chinese rhesus macaque specific pathogen-free (SPF) population using single nucleotide polymorphic (SNP) markers, by S. Kanthaswamy, J. Satkoski, A. Kou, V. Malladi, & D. Glenn Smith; Abstracts of oral presentations and poster session of the 27th Annual Symposium on Non-human Primate Models for AIDS, held in Boston, Massachusetts, on October 28�31, 2009.
* Tuberculosis & Malaria. Science, 2010, 328[5980], <www.sciencemag.org/content/vol328/issue5980>.
* Sexual dimorphism in primate aerobic capacity: A phylogenetic test. P. Lindenfors, L. J. Revell, & C. L. Nunn (Dept of Zoology, Stockholm University, S-106 91 Stockholm, Sweden [e-mail: [email protected]]). Journal of Evolutionary Biology, 2010, 23, 1183-1194, <www3.interscience.wiley.com/journal/123341243/abstract>.
. . . �Male intrasexual competition should favor increased male physical prowess. This should in turn result in greater aerobic capacity in males than in females (i.e. sexual dimorphism) and a correlation between sexual dimorphism in aerobic capacity and the strength of sexual selection among species. However, physiological scaling laws predict that aerobic capacity should be lower per unit body mass in larger than in smaller animals, potentially reducing or reversing the sex difference and its association with measures of sexual selection. We used measures of hematocrit and red blood cell (RBC) counts from 45 species of primates to test four predictions related to sexual selection and body mass: (i) on average, males should have higher aerobic capacity than females, (ii) aerobic capacity should be higher in adult than juvenile males, (iii) aerobic capacity should increase with increasing sexual selection, but also that (iv) measures of aerobic capacity should co-vary negatively with body mass. For the first two predictions, we used a phylogenetic paired t-test developed for this study. We found support for predictions (i) and (ii). For prediction (iii), however, we found a negative correlation between the degree of sexual selection and aerobic capacity, which was opposite to our prediction. Prediction (iv) was generally supported. We also investigated whether substrate use, basal metabolic rate, and agility influenced physiological measures of oxygen transport, but we found only weak evidence for a correlation between RBC count and agility.�
* Honest olfactory ornamentation in a female-dominant primate. Boulet, M., Crawford, J. C., Charpentier, M. J. E., & Drea, C. M. (C. M. D., Dept of Evol. Anthropology, Duke Univ., 129 Biol. Sci. Bldg., Science Dr., Box 90383, Durham, NC 27708-0383 [e-mail: [email protected]]. Journal of Evolutionary Biology, 2010, 23, 1558-1563, <www3.interscience.wiley.com/cgi-bin/fulltext/123443721/PDFSTART>.
. . . �Sexual selection theory predicts that potential mates or competitors signal their quality to conspecifics. Whereas evidence of honest visual or vocal signals in males abounds, evidence of honest signalling via scent or by females is scarce. We previously showed that scent marks in male lemurs seasonally encode information about individual heterozygosity � a reliable predictor of immunocompetence and survivorship. As female lemurs dominate males, compete over resources, and produce sexually differentiated scent marks that likely evolved via direct selection, here we tested whether females also advertise genetic quality via olfactory cues. During the breeding season specifically, individual heterozygosity correlated negatively with the diversity of fatty acids (FAs) expressed in labial secretions and positively with the diversity of heavy FA esters. As odor�gene relationships predictive of health and survivorship emerged during a period critical to mate choice and female competition, we posit that genital scent marks function as honest olfactory ornaments in females.�
* Mapping primary gyrogenesis during fetal development in primate brains: High-resolution in utero structural MRI study of fetal brain development in pregnant baboons. Kochunov, P., Castro, C., Davis, D., Dudley, D., Brewer, J., Zhang, Y., Kroenke, C. D., Purdy, D., Fox, P. T., Simerly, C., & Schatten, G. (Research Imaging Inst., Univ. of Texas Health Sci. Cntr, San Antonio, TX 78229 [e-mail: [email protected]]). Frontiers in Neurogenesis, 2010, 4[20], <frontiersin.org/neurogenesis/10.3389/fnins.2010.00020/abstract>.
. . . �The global and regional changes in the fetal cerebral cortex in primates were mapped during primary gyrification (PG; weeks 17-25 of 26 weeks total gestation). Studying pregnant baboons using high-resolution MRI in utero, measurements included cerebral volume, cortical surface area, gyrification index and length and depth of ten primary cortical sulci. Seven normally developing fetuses were imaged in two animals longitudinally and sequentially. We compared these results to those on PG that from the ferret studies and analyzed them in the context of our recent studies of phylogenetics of cerebral gyrification. We observed that in both primates and non-primates, the cerebrum undergoes a very rapid transformation into the gyrencephalic state, subsequently accompanied by an accelerated growth in brain volume and cortical surface area. However, PG trends in baboons exhibited some critical differences from those observed in ferrets. For example, in baboons, the growth along the long (length) axis of cortical sulci was unrelated to the growth along the short (depth) axis and far outpaced it. Additionally, the correlation between the rate of growth along the short sulcal axis and heritability of sulcal depth was negative and approached significance (r = -0.60; p < 0.10), while the same trend for long axis was positive and not significant (p = 0.3; p = 0.40). These findings, in an animal that shares a highly orchestrated pattern of PG with humans, suggest that ontogenic processes that influence changes in sulcal length and depth are diverse and possibly driven by different factors in primates than in non-primates.�
* Comparison of neural activity related to working memory in primate dorsolateral prefrontal and posterior parietal cortex. Qi, X., Katsuki, F., Meyer, T., Rawley, J. B., Zhou, X., Douglas, K. L., & Constantinidis, C. (C. C., Dept of Neurobiol. & Anat., Wake Forest Univ. Sch. of Med., Winston-Salem, NC 27157-1010 [e-mail: cconstan@ wfubmc.edu]). Frontiers in Systems Neuroscience, 2010, 4[12], <www.ncbi.nlm.nih.gov/pubmed/20514341>.
. . . �Neurons in a distributed network of cortical and subcortical areas continue to discharge after the presentation and disappearance of stimuli, providing a neural correlate for working memory. While it is thought that the prefrontal cortex plays a central role in this network, the relative contributions of other brain areas are not as well understood. In order to compare the contributions of the dorsolateral prefrontal and posterior parietal cortex, we recorded neurophysiological activity in monkeys trained to perform two different visuo-spatial working memory tasks: a Match/Nonmatch task, and a Spatial Delayed-Match-to-Sample Task. Neurons in both areas exhibited discharges in the delay periods of the tasks that could be classified in two forms. Sustained discharges persisted after the presentation of a stimulus in the receptive field with a constant or declining rate. Anticipatory responses increased in rate during the delay period, often appearing after presentation of a stimulus out of the receptive field. Despite similarities, we uncovered distinct differences between patterns of delay period in each brain area. Only in the prefrontal cortex sustained responses related to the original stimulus survived presentation of a second stimulus, in the context of the Match/Nonmatch task. Our results provide insights on the nature of processing in two areas active during working memory, and on the unique role of the prefrontal cortex in memory maintenance.�
* Orientation and direction-of-motion response in the middle temporal visual area (MT) of New World owl monkeys as revealed by intrinsic-signal optical imaging. Kaskan, P. M., Dillenburger, B. C., Lu, H. D., Roe, A. W., & Kaas, J. H. (J. H. K., Dept of Psych., Vanderbilt Univ., 301 Wilson Hall, 111 21st Ave South, Nashville, TN 37203 [e-mail: [email protected]]). Frontiers in Neuroenergetics, 2010, 4[23], <frontiersin.org/neuroanatomy/10.3389/fnana.2010.00023/full>.
. . .Intrinsic-signal optical imaging was used to evaluate relationships of domains of neurons in visual area MT selective for stimulus orientation and direction of motion. Maps of activation were elicited in MT of owl monkeys by gratings drifting back-and-forth, flashed stationary gratings and unidirectionally drifting fields of random dots. Drifting gratings, typically used to reveal orientation preference domains, contain a motion component that may be represented in MT. Consequently, this stimulus could activate groups of cells responsive to the motion of the grating, its orientation or a combination of both. Domains elicited from either moving or static gratings were remarkably similar, indicating that these groups of cells are responding to orientation, although they may also encode information about motion. To assess the relationship between domains defined by drifting oriented gratings and those responsive to direction of motion, the response to drifting fields of random dots was measured within domains defined from thresholded maps of activation elicited by the drifting gratings. The optical response elicited by drifting fields of random dots was maximal in a direction orthogonal to the map of orientation preference. Thus, neurons in domains selective for stimulus orientation are also selective for motion orthogonal to the preferred stimulus orientation.
* Metabolic adaptation for low energy throughput in orangutans. Pontzer, H., Raichlen, D. A., Shumaker, R. W., Ocobock, C., & Wich, S. A. (Dept of Anthropology, Washington Univ., St Louis, MO 63130 [e-mail: [email protected]]). Proceedings of the National Academy of Sciences, U.S.A. , 2010, 107, 14048-14052, <www.pnas.org/content/107/32/14048.full>.
. . . �Energy is the fundamental currency of life�needed for growth, repair, and reproduction�but little is known about the metabolic physiology and evolved energy use strategies of the great apes, our closest evolutionary relatives. Here we report daily energy use in free-living orangutans (Pongo spp.) and test whether observed differences in energy expenditure among orangutans, humans, and other mammals reflect known differences in life history. Using the doubly labeled water method, we measured daily energy expenditure (kCal/d) in orangutans living in a large indoor/outdoor habitat at the Great Ape Trust. Despite activity levels similar to orangutans in the wild, Great Ape Trust orangutans used less energy, relative to body mass, than nearly any eutherian mammal ever measured, including sedentary humans. Such an extremely low rate of energy use has not been observed previously in primates, but is consistent with the slow growth and low rate of reproduction in orangutans, and may be an evolutionary response to severe food shortages in their native Southeast Asian rainforests. These results hold important implications for the management of orangutan populations in captivity and in the wild, and underscore the flexibility and interdependence of physiological, behavioral, and life history strategies in the evolution of apes and humans.�
* Stress coping stimulates hippocampal neurogenesis in adult monkeys. Lyons, D. M., Buckmaster, P. S., Lee, A. G., Wu, C., Mitra, R., Duffey, L. M., Buckmaster, C. L., Her, S., Patel, P. D., & Schatzberg, A. F. (Dept of Psychiatry & Behavioral Sci., Stanford Univ. Med. Ctr, Stanford, CA 94305 [e-mail: [email protected]]). Proceedings of the National Academy of Sciences, U.S.A. , 2010, 107, 14823-14827, <www.pnas.org/content/107/33/14823.full>.
. . . �Coping with intermittent social stress is an essential aspect of living in complex social environments. Coping tends to counteract the deleterious effects of stress and is thought to induce neuroadaptations in corticolimbic brain systems. Here we test this hypothesis in adult squirrel monkey males exposed to intermittent social separations and new pair formations. These manipulations simulate conditions that typically occur in male social associations because of competition for limited access to residency in mixed-sex groups. As evidence of coping, we previously confirmed that cortisol levels initially increase and then are restored to prestress levels within several days of each separation and new pair formation. Follow-up studies with exogenous cortisol further established that feedback regulation of the hypothalamic-pituitary-adrenal axis is not impaired. Now we report that exposure to intermittent social separations and new pair formations increased hippocampal neurogenesis in squirrel monkey males. Hippocampal neurogenesis in rodents contributes to spatial learning performance, and in monkeys we found that spatial learning was enhanced in conditions that increased hippocampal neurogenesis. Corresponding changes were discerned in the expression of genes involved in survival and integration of adult-born granule cells into hippocampal neural circuits. These findings support recent indications that stress coping stimulates hippocampal neurogenesis in adult rodents. Psychotherapies designed to promote stress coping potentially have similar effects in humans with major depression.�
* Paired neuron recordings in the prefrontal and inferotemporal cortices reveal that spatial selection precedes object identification during visual search. Monosov, I. E., Sheinberg, D. L., & Thompson, K. G. (Lab. of Sensor. Res., NEI, NIH, Bethesda, MD 20892 [e-mail: [email protected]]). Proceedings of the National Academy of Sciences, U.S.A. , 2010, 29, 13105-13110, <www.pnas.org/content/107/29/13105.full>.
. . . �We addressed the question of how we locate and identify objects in complex natural environments by simultaneously recording single neurons from two brain regions that play different roles in this familiar activity�the frontal eye field (FEF), an area in the prefrontal cortex that is involved in visual spatial selection, and the inferotemporal cortex (IT), which is involved in object recognition�in monkeys performing a covert visual search task. Although the monkeys reported object identity, not location, neural activity specifying target location was evident in FEF before neural activity specifying target identity in IT. These two distinct processes were temporally correlated implying a functional linkage between the end stages of �where� and �what� visual processing and indicating that spatial selection is necessary for the formation of complex object representations associated with visual perception.�
* Receptive field asymmetries produce color-dependent direction selectivity in primate lateral geniculate nucleus. Tailby, C., Dobbie, W. J., Solomon, S. G., Szmajda, B. A., Hashemi-Nezhad, M., Forte, J. D., & Martin, P. R. (P. R. M., Univ. of Sydney, Eye Hospital Campus, Macquarie St., GPO Box 4337, Sydney NSW 2001, Australia [e-mail: [email protected]]. Journal of Vision, 2010, 10[8], <www.journalofvision.org/content/10/8/1.full>.
. . . �Blue-on receptive fields recorded in primate retina and lateral geniculate nucleus are customarily described as showing overlapping blue-on and yellow-off receptive field components. However, the retinal pathways feeding the blue-on and yellow-off subfields arise from spatially discrete receptor populations, and recent studies have given contradictory accounts of receptive field structure of blue-on cells. Here we analyzed responses of blue-on cells to drifting gratings, in single-cell extracellular recordings from the dorsal lateral geniculate nucleus in marmosets. We show that most blue-on cells exhibit selectivity for the drift direction of achromatic gratings. The standard concentric difference-of-Gaussians (DOG) model thus cannot account for responses of these cells. We apply a simple, anatomically plausible, extension of the DOG model. The model incorporates temporally offset elliptical two-dimensional Gaussian subfields. The model can predict color-contingent direction and spatial tuning. Because direction tuning in blue-on cells depends on stimulus chromaticity, spatial frequency, and temporal frequency, this property is of little value as a general mechanism for image movement detection. It is possible that anatomical wiring for color selectivity has constrained the capacity of blue-on cells to contribute to spatial and motion vision.�
* Corticomuscular coherence between motor cortex, somatosensory areas and forearm muscles in the monkey. Witham, C. L., Wang, M., & Baker, S. (S. B., Inst of Neurosci., Newcastle Univ., Newcastle upon Tyne, U.K.). Frontiers in System Neuroscience, 2010, 4[38], <www.frontiersin.org/systems%20neuroscience/10.3389/fnsys.2010.00038/abstract>.
. . . �Corticomuscular coherence has previously been reported between primary motor cortex (M1) and contralateral muscles. We examined whether such coherence could also be seen from somatosensory areas. Local field potentials (LFPs) were recorded from primary somatosensory cortex (S1; areas 3a and 2) and posterior parietal cortex (PPC) area 5 simultaneously with M1 LFP and forearm EMG activity in two monkeys during an index finger flexion task. Significant beta-band (~20 Hz) corticomuscular coherence was found in all areas investigated. Directed coherence (Granger causality) analysis was used to investigate the direction of effects. Surprisingly, the strongest beta-band directed coherence was in the direction from S1/PPC to muscle; it was much weaker in the ascending direction. Examination of the phase of directed coherence provided estimates of the time delay from cortex to muscle. Delays were longer from M1 (~62 ms for the first dorsal interosseous muscle) than from S1/PPC (~36 ms). We then looked at coherence and directed coherence between M1 and S1 for clues to this discrepancy. Directed coherence showed large beta-band effects from S1/PPC to M1, with smaller directed coherence in the reverse direction. The directed coherence phase suggested a delay of ~40 ms from M1 to S1. Corticomuscular coherence from S1/PPC could involve multiple pathways; the most important is probably common input from M1 to S1/PPC and muscles. If correct, this implies that somatosensory cortex receives oscillatory efference copy information from M1 about the motor command. This could allow sensory inflow to be interpreted in the light of its motor context.�
* Decoding complete reach and grasp actions from local primary motor cortex populations. Vargas-Irwin, C. E., Shakhnarovich, G., Yadollahpour, P., Mislow, J. M. K., Black, M. J., & Donoghue, J. P. (Dept of Neurosci., Brown Univ., Box G-LN, Providence, RI 02912 [e-mail: [email protected]]). The Journal of Neuroscience, 2010, 30, 9659-9669, <www.jneurosci.org/cgi/content/full/30/29/9659>.
. . . �How the activity of populations of cortical neurons generates coordinated multijoint actions of the arm, wrist, and hand is poorly understood. This study combined multielectrode recording techniques with full arm motion capture to relate neural activity in primary motor cortex (M1) of macaques (Macaca mulatta) to arm, wrist, and hand postures during movement. We find that the firing rate of individual M1 neurons is typically modulated by the kinematics of multiple joints and that small, local ensembles of M1 neurons contain sufficient information to reconstruct 25 measured joint angles (representing an estimated 10 functionally independent degrees of freedom). Beyond showing that the spiking patterns of local M1 ensembles represent a rich set of naturalistic movements involving the entire upper limb, the results also suggest that achieving high-dimensional reach and grasp actions with neuroprosthetic devices may be possible using small intracortical arrays like those already being tested in human pilot clinical trials.�
* Identification of residues within the L2 region of rhesus TRIM5α that are required for retroviral restriction and cytoplasmic body localization. Sastri, J., O�Connor, C., Danielson, C. M., McRaven, M., Perez, P., Diaz-Griffero, F., & Campbell, E. M. (E. M. C., 2160 S. First Ave, Maywood, IL 60153 [e-mail: [email protected]]. Virology, 2010, 405, 259-266.
. . . �The intracellular restriction factor TRIM5α inhibits infection by numerous retroviruses in a species-specific manner. The best characterized example of this restriction is the TRIM5α protein from rhesus macaques (rhTRIM5α), which potently inhibits HIV-1 infection. TRIM5α localizes to cytoplasmic assemblies of protein referred to as cytoplasmic bodies, though the role that these bodies play in retroviral restriction is unclear. We employed a series of truncation mutants to identify a discrete region, located within the Linker2 region connecting the coiled-coil and B30.2/PRYSPRY domains of TRIM5α, which is required for cytoplasmic body localization. Deletion of this region in the context of full-length rhTRIM5α abrogates cytoplasmic body localization. Alanine mutagenesis of the residues in this region identifies two stretches of amino acids that are required for both cytoplasmic body localization and retroviral restriction. This work suggests that the determinants that mediate TRIM5α localization to cytoplasmic bodies play a requisite role in retroviral restriction.�
* Comparison of lactate, base excess, bicarbonate, and pH as predictors of mortality after severe trauma in rhesus macaques (Macaca mulatta). Hobbs, T. R., O�Malley, J. P., Khouangsathiene, S., & Dubay, C. J. (Oregon NPRC, Oregon Health and Sciences Univ., Beaverton, OR 97006 [e-mail: [email protected]]). Comparative Medicine, 2010, 60, 233-239, <aalas.publisher.ingentaconnect.com/content/aalas/cm>.
. . . �Social group housing of rhesus macaques at biomedical facilities is advocated to improve the psychologic wellbeing of these intelligent and social animals. An unintended outcome of social housing in this species is increased intraspecific aggression resulting in cases of severe multiple trauma and posttraumatic shock. The metabolic correlates of oxygen debt are likely important quantifiers of the severity of posttraumatic shock and may serve as useful guides in the treatment of these cases. The purpose of this retrospective study was to evaluate venous blood lactate, base excess, bicarbonate, and pH as predictors of mortality. These four variables were assessed in 84 monkeys with severe traumatic injury and shock. Data were available from blood samples collected prior to resuscitation therapy and the day after resuscitation therapy. The pre- and postresuscitation therapy levels of the variables then were tested for association with 6-day survival. When measured prior to resuscitation therapy, all variables were strongly correlated with each other and had a statistically significant association with survival. No single variable had both strong specificity and high sensitivity when measured prior to resuscitation therapy. Survival analysis showed that as the number of categorical indicators of acidosis increased, 6-day survival decreased. Analysis of the four variables after resuscitation therapy indicated that lactate was the only variable significantly associated with survival in our study.�
* Contingent cooperation between wild female baboons. Cheney, D. L., Moscovice, L. R., Heesen, M., Mundry, R., & Seyfarth, R. M. (Dept of Biol., Univ. of Pennsylvania, Philadelphia, PA 19104 [e-mail: [email protected]]). Proceedings of the National Academy of Sciences, U.S.A. , 2010, 107, 9562-9566, <www.pnas.org/content/107/21/9562.full>.
. . . �The apparent rarity of contingent cooperation in animals has convinced many investigators that such reciprocity is unimportant, stimulating consideration of alternative explanations for cooperation, such as by-product mutualism and biological markets motivated by the likelihood of immediate reward. Nevertheless, there is also limited evidence that animals do sometimes rely on memory of recent interactions when behaving altruistically toward others. Here we describe a playback experiment conducted on wild female baboons, suggesting that contingent cooperation may occur among unrelated individuals, even when there is a temporal delay between the two cooperative acts. Hearing the recruitment call of an unrelated recent grooming partner caused subjects to move in the direction of the loudspeaker and approach their former partner, particularly when this partner had an infant. When the subject and her partner were close kin no such effect was observed. Subjects� responses were not influenced by any type of recent interaction, because prior grooming and prior aggression influenced their behavior in qualitatively different ways. Similarly, their responses were not prompted only by the motivation to resume friendly interactions, because prior grooming alone did not elicit approach. Instead, subjects were most likely to approach their former grooming partner when they had also heard her recruitment call. Results suggest that at least some forms of cooperation in animals may be based on memory of specific recent interactions.�
* The effects of four nursery rearing strategies on infant behavioral development in rhesus macaques (Macaca mulatta). Rommeck, I., Gottlieb, D. H., Strand, S. C., & McCowan, B. (California NPRC, U.C. Davis, CA 95616 [e-mail: [email protected]]). Journal of the American Association for Laboratory Animal Science, 2009, 48, 395-401, <www.ncbi.nlm.nih.gov/pubmed/19653949>.
. . . �Nursery rearing is the single most important risk factor in the development of severe forms of abnormal behavior, such as self-biting, in rhesus macaques. This practice is common in research laboratories and typically involves continuous pair housing of infants without maternal contact. We examined the effects of variation in peer socialization on the behavioral development of rhesus infants by exposing 32 newborn infants to four different socialization routines: continuously paired; intermittently paired; continuously paired rotationally (partners rotated within the group once a week); and intermittently paired rotationally. Analyses revealed that infants paired intermittently exhibited �floating limb� and self-biting behavior at significantly higher frequencies than those reared by using any other strategy. Results also suggested that continuous pairing was most effective in reducing the development of abnormal behaviors (that is, self-bite and floating limb), whereas intermittent pairing significantly reduced partner clinging and geckering. A principal component analysis revealed that floating limb behavior and self-biting are strongly associated. Self-biting began as early as 32 days of age, and a negative binomial regression on data of floating limb and self-biting revealed that early development of floating limb behavior predicts self-biting behavior later in development. Despite the significant effects of rearing strategies on the frequency of abnormal behaviors, we note that animals in all four treatment groups developed these traits to some degree. We suspect that the solitary incubator environment may be a trigger for the development of abnormal behaviors.�
* Consolation as possible expression of sympathetic concern among chimpanzees. Romero, T., Castellanos, M. A., & de Waal, F. B. M. (Living Links, Yerkes NPRC, Emory Univ., Atlanta, GA 30322 [e-mail: [email protected]]). Proceedings of the National Academy of Sciences, U.S.A. , 2010, 27, 12110-12115, <www.pnas.org/content/107/27/12110.full>.
. . .Chimpanzees are known to spontaneously provide contact comfort to recent victims of aggression, a behavior known as consolation. Similar behavior in human children is attributed to empathic or sympathetic concern. In line with this empathy hypothesis, chimpanzee consolation has been shown to reduce the recipient�s state of arousal, hence to likely alleviate distress. Other predictions from the empathy hypothesis have rarely been tested, however, owing to small sample sizes in previous studies. An exceptionally large database of spontaneous consolation in two outdoor-housed groups of chimpanzees lends further support to the empathy hypothesis in that consolation occurred disproportionally between individuals that are socially close (i.e., kin and affiliation partners) and was more typical of females than males, which differences are also known of human empathy. These effects were demonstrated using generalized linear mixed models, which control multiple variables at once. An exception to the above pattern was formed by the highest-ranking males, which frequently offered consolation to victims of aggression, probably as part of their general policing function in chimpanzee society. Consolation occurred more frequently in the absence of reconciliation between former opponents, suggesting that actors are sensitive to the contact need of victims of aggression, which may be greater if the aggressor ignores them. That consolation is an integrated part of close mutual relationships is supported by the tendency for it being reciprocated.
* Stand structure of a primate rich rainforest region in the central Western Ghats of southern India. Roy, K., Singh, M., Sushma, H. S., & Singh, M. (Biopsych. Lab., Univ. of Mysore, Manasagangotri, Mysore, Karnataka 570006, India [e-mail: [email protected]]). Journal of Threatened Taxa, 2010, 2, 930-939, <www.threatenedtaxa.org>.
. . . �The Western Ghats of southern India are one of the most important biodiversity regions in the world, not only due to their faunal diversity and abundance but also due to different habitat types, floral diversity and the presence of several endemic plant species. The rainforests in the central Western Ghats are inhabited by several primate species. We investigated the vegetation pattern and tree species occupancy of one of the prime primate habitats in the central Western Ghats. Lion-tailed macaque (Macaca silenus), bonnet macaque (Macaca radiata), Hanuman langur (Semnopithecus entellus achates), and Malabar slender loris (Loris lydekkerianus malabaricus) inhabit the study area. We studied the density, dominance, frequency and Importance Value Index (IVI) of different tree species, using the belt transect method on randomly selected plots covering 4.1ha. We found that all the plant species that were the most dominant species with high IVI in the forest were also used by the diurnal primates for foraging. Knema attenuata and Syzygium gardneri were found to be the �keystone� species. Since the forests of the study area do not come under the �protected area network� for wildlife, the data obtained during this study will be helpful in the forestry management practices with a view for wildlife conservation of the region.�
* International population movements and regional Plasmodium falciparum malaria elimination strategies. Tatem, A. J., & Smith, D. L. (Emerging Pathogens Inst., Univ. of Florida, Gainesville, FL 32610 [e-mail: [email protected]]). Proceedings of the National Academy of Sciences, U.S.A. , 2010, 27, 12222-12227, <www.pnas.org/content/107/27/12222.full>.
. . .Calls for the eradication of malaria require the development of global and regional strategies based on a strong and consistent evidence base. Evidence from the previous global malaria eradication program and more recent transborder control campaigns have shown the importance of accounting for human movement in introducing infections to areas targeted for elimination. Here, census-based migration data were analyzed with network analysis tools, Plasmodium falciparum malaria transmission maps, and global population databases to map globally communities of countries linked by relatively high levels of infection movements. The likely principal sources and destinations of imported cases in each region were also mapped. Results indicate that certain groups of countries, such as those in West Africa and central Asia, are much more strongly connected by relatively high levels of population and infection movement than others. In contrast, countries such as Ethiopia and Myanmar display significantly greater isolation in terms of likely infection movements in and out. The mapping here of both communities of countries linked by likely higher levels of infection movement, and �natural� migration boundaries that display reduced movement of people and infections between regions has practical utility. These maps can inform the design of malaria elimination strategies by identifying regional communities of countries afforded protection from recolonization by surrounding regions of reduced migration. For more isolated countries, a nationally focused control or elimination program is likely to stand a better chance of success than those receiving high levels of visitors and migrants from high-transmission regions.
* Postexposure treatment of Marburg virus infection. Geisbert, T. W., Hensley, L. E., Geisbert, J. B., Leung, A., Johnson, J. C., Grolla, A., & Feldmann, H. (Boston Univ. School of Med., NEIDLI, 620 Albany St, Rm 401B, Boston, MA 02118 [e-mail: [email protected]]). Emerging Infectious Diseases, 2010, 16, <www.cdc.gov/EID/content/16/7/1119.htm>.
. . . �Rhesus monkeys are protected from disease when a recombinant vesicular stomatitis virus-based vaccine is administered 20�30 min after infection with Marburg virus. We protected 5/6 monkeys when this vaccine was given 24 h after challenge; 2/6 animals were protected when the vaccine was administered 48 h postinfection.�
* A fossil primate of uncertain affinities from the earliest late Eocene of Egypt. Seiffert, E. R., Simons, E. L., Boyer, D. M., Perry, J. M. G., Ryan, T. M., & Sallam, H. M. (Dept of Anatomical Sci., Stony Brook Univ., Stony Brook, NY 11794-8081 [e-mail: [email protected]]). Proceedings of the National Academy of Sciences, U.S.A. , 2010, 107, 9712-9717, <www.pnas.org/content/107/21/9712.full>.
. . . �Paleontological work carried out over the last 3 decades has established that three major primate groups were present in the Eocene of Africa�anthropoids, adapiforms, and advanced strepsirrhines. Here we describe isolated teeth of a previously undocumented primate from the earliest late Eocene (≈37 Ma) of northern Egypt, Nosmips aenigmaticus, whose phylogenetic placement within Primates is unclear. Nosmips is smaller than the sympatric adapiform Afradapis but is considerably larger than other primate taxa known from the same paleocommunity. The species bears an odd mosaic of dental features, combining enlarged, elongate, and molariform premolars with simple upper molars that lack hypocones. Phylogenetic analysis across a series of different assumption sets variously places Nosmips as a stem anthropoid, a nonadapiform stem strepsirrhine, or even among adapiforms. This phylogenetic instability suggests to us that Nosmips likely represents a highly specialized member of a previously undocumented, and presumably quite ancient, endemic African primate lineage, the subordinal affinities of which have been obscured by its striking dental autapomorphies. Discriminant functions based on measurements of lower molar size and topography reliably classify extant prosimian primates into their correct dietary groups and identify Nosmips and Afradapis as omnivores and folivores, respectively. Although Nosmips currently defies classification, this strange and unexpected fossil primate nevertheless provides additional evidence for high primate diversity in northern Africa ≈37 million years ago and further underscores the fact that our understanding of early primate evolution on that continent remains highly incomplete.�
* A draft sequence of the Neandertal genome. Green, R. E., Krause, J., Briggs, A. W., Maricic, T., Stenzel, U., Kircher, M., Patterson, N., Li, H., Zhai, W., Fritz, M. H.-Y., Hansen, N. F., Durand, E. Y., Malaspinas, A.-S., Jensen, J. D., Marques-Bonet, T., Alkan, C., Pr�fer, K., Meyer, M., Burbano, H. A., Good, J. M., Schultz, R., Aximu-Petri, A., Butthof, A., H�ber, B., H�ffner, B., Siegemund, M., Weihmann, A., Nusbaum, C., Lander, E. S., Russ, C., Novod, N., Affourtit, J., Egholm, M., Verna, C., Rudan, P., Brajkovic, D., Kucan, Z., Gusic, I., Doronichev, V. B., Golovanova, L. V., Lalueza-Fox, C., de la Rasilla, M., Fortea, J., Rosas, A., Schmitz, R. W., Johnson, P. L. F., Eichler, E. E., Falush, D., Birney, E.,. Mullikin, J. C., Slatkin, M., Nielsen, R., Kelso, J., Lachmann, M., Reich, D., & P��bo, S. (Dept of Evol. Genetics, Max-Planck Inst. for Evol. Anthrop., D-04103 Leipzig, Germany [e-mail: [email protected]] ). Science, 2010, 328, 710-722.
. . . �Neandertals, the closest evolutionary relatives of present-day humans, lived in large parts of Europe and western Asia before disappearing 30,000 years ago. We present a draft sequence of the Neandertal genome composed of more than 4 billion nucleotides from three individuals. Comparisons of the Neandertal genome to the genomes of five present-day humans from different parts of the world identify a number of genomic regions that may have been affected by positive selection in ancestral modern humans, including genes involved in metabolism and in cognitive and skeletal development. We show that Neandertals shared more genetic variants with present-day humans in Eurasia than with present-day humans in sub-Saharan Africa, suggesting that gene flow from Neandertals into the ancestors of non-Africans occurred before the divergence of Eurasian groups from each other.�
* Targeted investigation of the Neandertal genome by array-based sequence capture. Burbano, H. A., Hodges, E., Green, R. E., Briggs, A. W., Krause, J., Meyer, M., Good, J. M., Maricic, T., Johnson, P. L. F., Xuan, Z., Rooks, M., Bhattacharjee, A., Brizuela, L., Albert, F. W., de la Rasilla, M., Fortea, J., Rosas, A., Lachmann, M., Hannon, G. J., & P��bo, S. (Max Planck Inst. for Evol. Anthrop., D-04103 Leipzig, Germany). Science, 2010, 328, 723-725, <www.sciencemag.org/cgi/content/full/328/5979/723>.
. . . �It is now possible to perform whole-genome shotgun sequencing as well as capture of specific genomic regions for extinct organisms. However, targeted resequencing of large parts of nuclear genomes has yet to be demonstrated for ancient DNA. Here we show that hybridization capture on microarrays can successfully recover more than a megabase of target regions from Neandertal DNA even in the presence of ~99.8% microbial DNA. Using this approach, we have sequenced ~14,000 protein-coding positions inferred to have changed on the human lineage since the last common ancestor shared with chimpanzees. By generating the sequence of one Neandertal and 50 present-day humans at these positions, we have identified 88 amino acid substitutions that have become fixed in humans since our divergence from the Neandertals.�
* Chimpanzee and human Y chromosomes are remarkably divergent in structure and gene content. Hughes, J. F., Skaletsky, H., Pyntikova, T., Graves, T. A., van Daalen, S. K. M., Minx, P. J., Fulton, R. S., McGrath, S. D., Locke, D. P., Friedman, C., Trask, B. J., Mardis, E. R., Warren, W. C., Repping, S., Rozen, S., Wilson, R. K., & Page, D. C. (D. C. P., Howard Hughes Medical Institute, Whitehead Institute, and Department of Biology, Massachusetts Institute of Technology, 9 Cambridge Center, Cambridge, Massachusetts 02142 [e-mail: [email protected]]). Nature, 2010, 463, 536-539, <www.nature.com/nature/journal/v463/n7280/full/nature08700.html>.
. . . �The human Y chromosome began to evolve from an autosome hundreds of millions of years ago, acquiring a sex-determining function and undergoing a series of inversions that suppressed crossing over with the X chromosome. Little is known about the recent evolution of the Y chromosome because only the human Y chromosome has been fully sequenced. Prevailing theories hold that Y chromosomes evolve by gene loss, the pace of which slows over time, eventually leading to a paucity of genes, and stasis. These theories have been buttressed by partial sequence data from newly emergent plant and animal Y chromosomes, but they have not been tested in older, highly evolved Y chromosomes such as that of humans. Here we finished sequencing of the male-specific region of the Y chromosome (MSY) in our closest living relative, the chimpanzee, achieving levels of accuracy and completion previously reached for the human MSY. By comparing the MSYs of the two species we show that they differ radically in sequence structure and gene content, indicating rapid evolution during the past 6 million years. The chimpanzee MSY contains twice as many massive palindromes as the human MSY, yet it has lost large fractions of the MSY protein-coding genes and gene families present in the last common ancestor. We suggest that the extraordinary divergence of the chimpanzee and human MSYs was driven by four synergistic factors: the prominent role of the MSY in sperm production, �genetic hitchhiking� effects in the absence of meiotic crossing over, frequent ectopic recombination within the MSY, and species differences in mating behavior. Although genetic decay may be the principal dynamic in the evolution of newly emergent Y chromosomes, wholesale renovation is the paramount theme in the continuing evolution of chimpanzee, human and perhaps other older MSYs.�
* A conserved pattern of brain scaling from sharks to primates. Yopak, K. E., Lisney, T. J., Darlington, R. B., Collin, S. P., Montgomery, J. C., & Finlay, B. L. (Ctr for Sci. Comput. Imaging, Univ. Calif., San Diego, CA 92037 [e-mail: [email protected]]). Proceedings of the National Academy of Sciences, U.S.A. , 2010, 29, 12946-12951, <www.pnas.org/content/107/29/12946.full>.
. . .Several patterns of brain allometry previously observed in mammals have been found to hold for sharks and related taxa (chondrichthyans) as well. In each clade, the relative size of brain parts, with the notable exception of the olfactory bulbs, is highly predictable from the total brain size. Compared with total brain mass, each part scales with a characteristic slope, which is highest for the telencephalon and cerebellum. In addition, cerebellar foliation reflects both absolute and relative cerebellar size, in a manner analogous to mammalian cortical gyrification. This conserved pattern of brain scaling suggests that the fundamental brain plan that evolved in early vertebrates permits appropriate scaling in response to a range of factors, including phylogeny and ecology, where neural mass may be added and subtracted without compromising basic function.
* New Oligocene primate from Saudi Arabia and the divergence of apes and Old World monkeys. Zalmout, I. S., Sanders, W. J., MacLatchy, L. M., Gunnell, G. F., Al-Mufarreh, Y. A., Ali, M. A., Nasser, A.-A. H., Al-Masari, A. M., Al-Sobhi, S. A., Nadhra, A. O., Matari, A. H., Wilson, J. A., & Gingerich, P. D. (Museum of Paleontology, Univ. of Michigan, 1109 Geddes Ave, Ann Arbor, MI 48109 [e-mail: [email protected]]). Nature, 2010, 466, 360-364, <www.nature.com/nature/journal/v466/n7304/full/nature09094.html>.
. . . �It is widely understood that Hominoidea (apes and humans) and Cercopithecoidea (Old World monkeys) have a common ancestry as Catarrhini deeply rooted in Afro-Arabia. The oldest stem Catarrhini in the fossil record are Propliopithecoidea, known from the late Eocene to early Oligocene epochs (roughly 35�30 Myr ago) of Egypt, Oman and possibly Angola. Genome-based estimates for divergence of hominoids and cercopithecoids range into the early Oligocene; however, the mid-to-late Oligocene interval from 30 to 23 Myr ago has yielded little fossil evidence documenting the morphology of the last common ancestor of hominoids and cercopithecoids, the timing of their divergence, or the relationship of early stem and crown catarrhines. Here we describe the partial cranium of a new medium-sized (about 15�20 kg) fossil catarrhine, Saadanius hijazensis, dated to 29�28 Myr ago. Comparative anatomy and cladistic analysis shows that Saadanius is an advanced stem catarrhine close to the base of the hominoid�cercopithecoid clade. Saadanius is important for assessing competing hypotheses about the ancestral morphotype for crown catarrhines, early catarrhine phylogeny and the age of hominoid�cercopithecoid divergence. Saadanius has a tubular ectotympanic but lacks synapomorphies of either group of crown Catarrhini, and we infer that the hominoid�cercopithecoid split happened later, between 29�28 and 24 Myr ago.�
* New immature hominin fossil from European Lower Pleistocene shows the earliest evidence of a modern human dental development pattern. Berm�dez de Castro, J. M., Martin�n-Torres, M., Prado, L., G�mez-Robles, A., Rosell, J., L�pez-Pol�n, L., Arsuaga, J. L., & Carbonell, E. (Centro Nac. de Investigaci�n sobre Evoluci�n Humana, 09002 Burgos, Spain [e-mail: [email protected]]). Proceedings of the National Academy of Sciences, U.S.A. , 2010, 107, 11739-11744, <www.pnas.org/content/107/26/11739>.
. . . �Here we present data concerning the pattern of dental development derived from the microcomputed tomography (microCT) study of a recently discovered immature hominin mandible with a mixed dentition recovered from the TD6 level of the Gran Dolina Lower Pleistocene cave site in Sierra de Atapuerca, northern Spain. These data confirm our previous results that nearly 1 million years ago at least one European hominin species had a fully modern pattern of dental development with a clear slowdown in the development of the molar field regarding the anterior dental field. Furthermore, using available information about enamel formation times and root extension rates in chimpanzees, early hominins, and modern humans, we have estimated that the formation time of the upper and lower first molars of individual 5 (H5) from TD6, which had just erupted at the time of the death of this individual, ranges between 5.3 and 6.6 y. Therefore, the eruption time of the first permanent molars (M1) in the TD6 hominins was within the range of variation of modern human populations. Because the time of M1 eruption in primates is a robust marker of life history, we suggest, as a working hypothesis, that these hominins had a prolonged childhood in the range of the variation of modern humans. If this hypothesis is true, it implies that the appearance in Homo of this important developmental biological feature and an associated increase in brain size preceded the development of the neocortical areas leading to the cognitive capabilities that are thought to be exclusive to Homo sapiens. �
* An early Australopithecus afarensis postcranium from Woranso-Mille, Ethiopia. Haile-Selassie, Y., Latimer, B. M., Alene, M., Deino, A. L., Gibert, L., Melillo, S. M., Saylor, B. Z., Scott, G. R., & Lovejoy, C. O. (Cleveland Museum of Natural History, Cleveland, OH 44106 [e-mail: [email protected]]). Proceedings of the National Academy of Sciences, U.S.A. , 2010, 27, 12121-12126, <www.pnas.org/content/107/27/12121.full>.
. . . �Only one partial skeleton that includes both forelimb and hindlimb elements has been reported for Australopithecus afarensis. The diminutive size of this specimen (A.L. 288-1 [�Lucy�]) has hampered our understanding of the paleobiology of this species absent the potential impact of allometry. Here we describe a large-bodied (i.e., well within the range of living Homo) specimen that, at 3.58 Ma, also substantially antedates A.L. 288-1. It provides fundamental evidence of limb proportions, thoracic form, and locomotor heritage in Australopithecus afarensis. Together, these characteristics further establish that bipedality in Australopithecus was highly evolved and that thoracic form differed substantially from that of either extant African ape.�
* Adenosine-to-inosine RNA editing shapes transcriptome diversity in primates. Paz-Yaacov, N., Levanon, E. Y., Nevo, E., Kinar, Y., Harmelin, A., Jacob-Hirsch, J., Amariglio, N., Eisenberg, E., & Rechavi, G. (G. R., Cancer Research Center, Chaim Sheba Medical Center, Tel Hashomer 52621, Israel [e-mail: [email protected]]). Proceedings of the National Academy of Sciences, U.S.A., 2010, 27, 12174-12179, <www.pnas.org/content/107/27/12174.full>.
. . . �Human and chimpanzee genomes are almost identical, yet humans express higher brain capabilities. Deciphering the basis for this superiority is a long sought-after challenge. Adenosine-to-inosine (A-to-I) RNA editing is a widespread modification of the transcriptome. The editing level in humans is significantly higher compared with nonprimates, due to exceptional editing within the primate-specific Alu sequences, but the global editing level of nonhuman primates has not been studied so far. Here we report the sequencing of transcribed Alu sequences in humans, chimpanzees, and rhesus monkeys. We found that, on average, the editing level in the transcripts analyzed is higher in human brain compared with nonhuman primates, even where the genomic Alu structure is unmodified. Correlated editing is observed for pairs and triplets of specific adenosines along the Alu sequences. Moreover, new editable species-specific Alu insertions, subsequent to the human�chimpanzee split, are significantly enriched in genes related to neuronal functions and neurological diseases. The enhanced editing level in the human brain and the association with neuronal functions both hint at the possible contribution of A-to-I editing to the development of higher brain function. We show here that combinatorial editing is the most significant contributor to the transcriptome repertoire and suggest that Alu editing adapted by natural selection may therefore serve as an alternate information mechanism based on the binary A/I code.�
* Use of biomarkers of Collagen Types I and III fibrosis metabolism to detect cardiovascular and renal disease in chimpanzees (Pan troglodytes). Ely, J. J., Bishop, M. A., Lammey, M. L., Sleeper, M. M., Steiner, J. M., & Lee, D. R. (Alamogordo Primate Facility, Charles River Consulting & Staffing Services, Holloman Airforce Base, NM 88330). Comparative Medicine, 2010, 60, 154-158, <www.ncbi.nlm.nih.gov/pubmed/20412692>.
. . . �Cardiovascular disease is the leading cause of morbidity and mortality among captive chimpanzees. The most prevalent form of cardiovascular disease among chimpanzees is sudden cardiac death. Myocardial fibrosis was the only significant pathologic lesion observed in affected animals at necropsy. We previously showed an association between myocardial fibrosis and sudden cardiac death. The presumed pathogenesis was interstitial myocardial fibrosis that led to decreased myocardial contractility and interrupted signal propagation in the heart, leading to fibrillation and resulting in sudden cardiac death. In this pilot study, we assayed 5 biomarkers of collagen types I and III metabolism and fibrogenesis and studied their association with CVD in chimpanzees. The biomarker MMP1 did not crossreact in chimpanzee sera and could not be studied further. Two biomarkers (TIMP1 and PINP) and their difference showed no significant association with CVD in chimpanzees. The biomarkers ICTP and PIIINP were significantly increased in cases of CVD with concurrent renal disease. Furthermore, both biomarkers showed a significant trend to increase with disease severity. We conclude that ICTP and PIIINP warrant further study for antemortem detection of renal and myocardial fibrosis in chimpanzees.�
* Differential changes in steroid hormones before competition in bonobos and chimpanzees. Wobber, V., Hare, B., Maboto, J., Lipson, S., Wrangham, R., & Ellison, P. T. (Dept of Human Evol. Biol., Harvard Univ., Peabody Museum, Cambridge, MA 02138 [e-mail: wobber@ fas.harvard.edu]). Proceedings of the National Academy of Sciences, U.S.A. , 2010, 107, 12457-12462, <www.pnas.org/content/107/28/12457.full>.
. . . �A large body of research has demonstrated that variation in competitive behavior across species and individuals is linked to variation in physiology. In particular, rapid changes in testosterone and cortisol during competition differ according to an individual�s or species� psychological and behavioral responses to competition. This suggests that among pairs of species in which there are behavioral differences in competition, there should also be differences in the endocrine shifts surrounding competition. We tested this hypothesis by presenting humans� closest living relatives, chimpanzees (Pan troglodytes) and bonobos (Pan paniscus), with a dyadic food competition and measuring their salivary testosterone and cortisol levels. Given that chimpanzees and bonobos differ markedly in their food-sharing behavior, we predicted that they would differ in their rapid endocrine shifts. We found that in both species, males showed an anticipatory decrease (relative to baseline) in steroids when placed with a partner in a situation in which the two individuals shared food, and an anticipatory increase when placed with a partner in a situation in which the dominant individual obtained more food. The species differed, however, in terms of which hormone was affected; in bonobo males the shifts occurred in cortisol, whereas in chimpanzee males the shifts occurred in testosterone. Thus, in anticipation of an identical competition, bonobo and chimpanzee males showed differential endocrine shifts, perhaps due to differences in perception of the situation, that is, viewing the event either as a stressor or a dominance contest. In turn, common selection pressures in human evolution may have acted on the psychology and the endocrinology of our competitive behavior.�
Contents of Volumes 46-48 (2007-2009)
* * *
All correspondence concerning the Newsletter should be addressed to:
Judith E. Schrier, Psychology Department, Box 1853, Brown University
Providence, Rhode Island 02912. [401-863-2511; fax: 401-863-1300]
e-mail address: Judith_Schrier@brown.edu
Current and back issues of the Newsletter are available on the World Wide Web at
http://www.brown.edu/primate
The Newsletter is supported by Brown University
Cover photograph of a white-handed gibbon(Hylobates lar), taken at the San Diego Zoo by Mark Abbott in 2007
Copyright (c) 2010 by Brown University
Last updated: September 17, 2010