 |
Office of Media Relations | |||
News | ||||
|
Neuroscience
Not So Different After All: Mysterious Eye Cells Adapt To Light
Unlike their cousins, rods and cones, newly discovered retinal cells don’t aid sight in a traditional sense. Instead, they constrict the eye’s pupil and set the body’s circadian clock. But new research from Brown University – where the photoreceptors were discovered by David Berson and colleagues – shows that these cells are sensitive to lighting conditions in a manner similar to rods and cones. Results appear in Neuron. PROVIDENCE, R.I. — A new retinal photoreceptor adjusts its sensitivity in different lighting conditions, according to scientists at Brown University, where the rare eye cells were discovered. The results, published in Neuron, are a surprise. Though rods and cones, their biological cousins in the retina, clearly adjust to light levels, these new cells – intrinsically photosensitive retinal ganglion cells, or ipRGCs – were assumed not to adapt this way. Still, the adaptation process in ipRGCs is weaker and slower than it is in rods and cones. 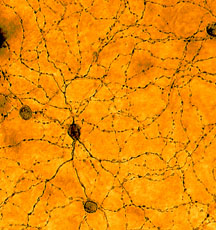 A complementary system in the eye The findings provide further evidence that the eye has complementary brain-signaling systems at work. Rods and cones rapidly communicate changes in brightness, signals which allow us to glimpse a baseball streaking across the sky or a deer darting into a darkened road. But ipRGCs work differently. They send signals about overall brightness, telling the brain when it is night and when it is day. “These cells operate like a light meter on a camera,” said David Berson, the Sidney A. Fox and Dorothea Doctors Fox Professor of Ophthalmology and Visual Sciences. “They tell the brain to constrict the pupil based on the amount of light registered over time.” Berson discovered ipRGCs in his Brown neuroscience lab three years ago. These cells number no more than 2,000 in the eye and have a direct link to the brain, sending electrical messages to an area regulating the pupil as well as a region controlling the body clock. This circadian rhythm controls alertness, sleep, hormone production, body temperature and organ function. Earlier this year, the Berson lab reported in Nature that a protein called melanopsin plays a key role in the cells’ ability to absorb light energy, sparking a chain reaction that sends these signals to the brain. But how ipRGCs behave naturally is largely unknown. The authors said this new work fills in a few blanks in the scientific book. “Until now, we didn’t know if these cells were adaptive to lighting conditions,” said Kwoon Wong, a postdoctoral research fellow in the Berson lab and the lead author of the Neuron paper. “Now we know that they are. Compared with rods and cones, they’re glacially slow and they don’t adjust their sensitivity as completely. But they do behave in similar ways. It’s a difference of degree, not of kind. In fact, what we found out about ipRGCs seems to be true of all biological systems: They adapt to their environment.” To arrive at the findings, Wong, Berson and former Brown undergraduate Felice Dunn exposed rat ipRGCs to a series of increasingly bright flashes then measured changes in voltage across the cell membrane – a sign of neuronal response. The flashes evoked much bigger responses when the cells had been sitting in darkness than when they were exposed to bright background light. These adjustments persisted when ipRGC synapses were chemically blocked, indicating that the changes in sensitivity were indeed happening within these cells. The National Institutes of Health funded the work. ###### Media Relations Home | Top of File | e-Subscribe | Brown Home Page | ||||