
|
May 15, 2007 |
Biomedical Engineering
Growing Nerve Cells in 3-D Dramatically Affects Gene Expression
Nerve cells grown in three-dimensional environments deploy hundreds of different genes compared with cells grown in standard two-dimensional petri dishes, according to a new Brown University study. The research, spearheaded by bioengineer Diane Hoffman-Kim, adds to a growing body of evidence that lab culture techniques dramatically affect the way these cells behave. | |||
|
Brown University Home |
PROVIDENCE, R.I. [Brown University] — When it comes to growing cells in a lab, technique matters. A new Brown University study shows that nerve cells grown in three-dimensional cultures use 1,766 genes differently compared to nerve cells grown in standard two-dimensional petri dishes. The study, published in the May issue of Tissue Engineering, adds to a growing body of research showing that culture techniques can significantly affect cell growth and function. This research shows that cells grown in a laboratory in 3-D environments, not in flat petri dishes, are more like cells grown in the ultimate 3-D environment – the human body. 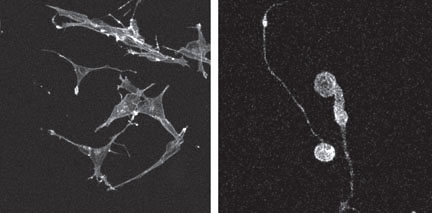 Dimensions of culture “More and more, we’re seeing evidence that cells cultured in three dimensions look and behave more like cells in your body,” said Diane Hoffman-Kim, the Brown bioengineer who spearheaded the new study, “so culture method is critical. If you want to better understand how the human body behaves or how new drugs might fight disease, 3-D may be a better bet.” For more than 100 years, scientists have grown human cells in flat dishes. In these 2-D glass incubators, better known as petri dishes, cells stick to the bottom and spread out as they multiply. But in the body, cells don’t grow that way. They are suspended in fluids and gels and surrounded by other cells. And these cells aren’t stuck; they move. As a result, some scientists suspect that hothouse cells do not behave like in vivo varieties. This means that the critical functions scientists are trying to understand by studying these cells – from the proliferation of cancer to the bacterial assault by antibiotics – may play out differently. Studies indeed show differences in behavior between cells cultured in 2-D and in 3-D. Cells cultured in 3-D, for example, grow faster. Hoffman-Kim, an assistant professor of medical science and engineering at Brown, wants to find a way to repair damaged nerve tissue. To get there, she needs to grow nerve cells that will flourish when placed inside the body. So Hoffman-Kim put 2-D and 3-D cell culture methods to a comparative test. Would the cells be different? Which ones would grow best? To answer that question, Hoffman-Kim and researchers in her lab took a line of cancerous nerve cells and cultured them in different environments. They placed one batch of cells in standard petri dishes coated with collagen. They suspended the other batch in a thicker collagen gel. The bioengineers took great pains to be sure that this one-dimension difference was the only difference in the culture conditions. Team members even counted individuals cells as they went into the dishes to be sure that each dish contained precisely the same amount. After a day of growth, the researchers extracted RNA from the cells and conducted a microarray analysis to determine differences in gene activity. The results: A whopping 1,766 genes responded differently, either switching on or switching off. To check their work, researchers repeated the experiments but this time produced multiple copies of eight genes specifically linked to cell growth. Those experiments confirmed the differences in gene activity. Hoffman-Kim and her team deployed high-powered microscopes to detect other differences. Cells cultured in 3-D had a rounder, more realistic shape, and their neurites – spindly projections sprouting from the cell body – were longer. “The cells in 3-D culture grew faster and looked more like the body’s cells,” she said. “This means that this culture method might create tissue that could be more successfully implanted.” Graduate student Grace Li and senior research assistant Liane Livi were the lead authors of the Tissue Engineering article. Graduate student Celinda Gourd and graduate alumna Elizabeth Deweerd also served on the research team. All are members of Brown’s Center for Biomedical Engineering and the Department of Molecular Pharmacology, Physiology, and Biotechnology in The Warren Alpert Medical School of Brown University. The National Center for Research Resources, part of the National Institutes of Health, funded the work through Brown University’s Center for Genetics and Genomics. Editors: Brown University has a fiber link television studio available for domestic and international live and taped interviews and maintains an ISDN line for radio interviews. For more information, call the Office of Media Relations at (401) 863-2476. ###### | |||