PERICARDIAL DISEASES
Pericardium
Anatomy (Table 1):The
normal pericardium is a fibroserous sac, which surrounds the heart and
adjoining portions of the great vessels. It has two layers. The inner visceral
layer, also known as the epicardium, consists of a thin layer of mesothelial
cells is closely adherent to the surface of the heart. It is reflected
onto the surface of the outer fibrous layer with which it forms the parietal
pericardium, which consists of collagenous fibrous tissue and elastic fibrils.
Between the two layers lies the pericardial space, which contains approximately
10-50ml of fluid, which is an ultrafiltrate of plasma. Drainage of pericardial
fluid is via right lymphatic duct and thoracic duct.
Functions of the Pericardium
(Table 2)
Although surgical removal of the pericardium (pericardiectomy)
and congenital absence of the pericardium are well tolerated conditions,
the pericardium is known to serve several functions. The three major functions
of the pericardium are:
1. Stabilization of the heart within the thoracic
cavity by virtue of its ligamentous attachments.
2. Protection of the heart from mechanical trauma
and infection from adjoining structures. The pericardial fluid functions
as a lubricant and decreases friction of cardiac surface during systole
and diastole.
3. Prevention of excessive dilation of heart especially
during sudden rise in intracardiac volume e.g. acute aortic or mitral regurgitation.
Intrapericardial pressure (IPP) tracks intrathoracic
pressure. Normally, with inspiration, the negative intrathoracic pressure
is transmitted to pericardial space and IPP drops. This results in an increase
in blood flow to the right ventricle. As a result of the latter, jugular
venous and right atrial pressures drop. The increase in right ventricular
volume causes the interventricular septum to shift towards the left ventricle
(LV) reducing left ventricular volume. This results in a drop in LV stroke
volume and therefore blood pressure. This drop in systolic blood pressure
during inspiration amounts to <10mmHg and is referred to as pulsus paradoxus.
A pulsus paradoxus in excess of 10mmHg is encountered in cardiac tamponade
(see Tamponade).
ETIOLOGY -- ACUTE PERICARDITIS
I. INFECTIVE
1. VIRAL - Coxsackie A and B, Influenza, adenovirus,
HIV, etc.
2. BACTERIAL - Staphylococcus, pneumococcus, tuberculosis,
etc.
3. FUNGAL - Candida
4. PARASITIC - Amoeba, candida, etc.
II. AUTOIMMUNE DISORDERS
1. Systemic lupus erythematosus (SLE), rheumatoid
arthritis, etc.
2. Drug-Induced lupus (e.g. Hydralazine, Procainamide)
3. Rheumatoid Arthritis
4. Post Cardiac Injury Syndromes i.e. postmyocardial
(Dressler's) Syndrome, postcardiotomy syndrome, etc.
III. NEOPLASM
1. Primary mesothelioma
2. Secondary, metastatic
3. Direct extension from adjoining tumor
IV. RADIATION PERICARDITIS
V. RENAL FAILURE (uremia)
VI. TRAUMATIC CARDIAC INJURY
1. Penetrating - stab wound, bullet wound
2. Blunt non-penetrating - automobile steering wheel
accident
VII. IDIOPATHIC
ETIOLOGY OF PERICARDITIS
Viral Pericarditis
Cardiotropic viruses - Coxsackie B, ECHO Type 8,
mumps, influenza, and infectious mono - may produce acute pericarditis.
These viruses may also be responsible for at least some of the cases of
"idiopathic" pericarditis. While HIV may cause pericarditis, most often
pericarditis in patients with AIDS is due to opportunistic infections e.g.
tuberculous, fungal or bacterial infection.
Pericarditis Due to Other Infective Agents
Pericarditis due to bacteria, fungi, or rickettsiae
is usually encountered in debilitated or immunocompromised patients or
as a complication of infection following thoracic surgery, pneumonia, abuse
of intravenous drugs, etc. Common bacterial agents include staphylococcus
aureus, pneumococcus, hemophilus influenza and, less commonly, gram-negative
rods.
Pericarditis Due to Immunologic/Connective
Tissue Diseases
(e.g. Lupus erythematosus)
Pericarditis often with effusion is encountered in
patients with systemic lupus erythematosus (SLE) and is more common
in young women. Drugs such as procainamide and hydralazine may cause a
lupus-like syndrome when used for a prolonged period.
Pericarditis of Myocardial Infarction
Q wave ("transmural") myocardial infarction may cause
acute pericarditis characterized by chest pain and pericardial rub within
24-48 hours of an infarction. The late-form, of pericarditis known
as post myocardial infarction syndrome or Dressler's syndrome occurs
2-4 weeks to several months after an infarction. It is characterized by
chest pain, pericardial rub, fever, high sedimentation rate, pleuritis,
pulmonary infiltrates, pericardial effusion and pleural effusion. The etiology
of this entity is unclear but is probably related to an autoimmune response
directed against antigens released by damaged myocardial cells. A related
condition, post-cardiotomy syndrome, is seen in some patients after
cardiac surgery. The term postcardiac injury syndrome is used to describe
both entities.
Neoplastic Pericarditis
Primary malignant pericardial disease (mesothelioma,
sarcoma) is very rare. More frequently, malignancy is secondary to a local
spread from an adjoining structure e.g. bronchogenic carcinoma or metastatic
spread via lymphatics or blood, e.g. breast cancer. Effusions are hemorrhagic
and tamponade in common. In some cases tumor encasement of the pericardium
leads to constrictive pericarditis.
Uremic Pericarditis
Uremia associated with chronic renal failure is a
common cause of pericarditis and pericardial effusion. The etiology of
pericardial effusion in these patients is unclear but may be related to
metabolic factors, infection, bleeding in the pericardial space from thrombocytopenia
or autoimmune disorder. The condition may progress to constrictive pericarditis
after months or years.
PATHOLOGY (Table 3)
Inflammation of the pericardium or pericarditis causes
deposition of fibrinous material over the inner lining of the pericardial
surfaces producing a shaggy or "bread and butter" appearance. The rubbing
of these surfaces during the cardiac cycle produces one of the classic
features of pericarditis i.e. pericardial rub. Inflammation also results
in the production of pericardial effusion with a high leukocyte count and,
depending on the etiology, one may detect the presence of tumor cells,
LE cells, bacteria, etc.
SIGNS AND SYMPTOMS
OF ACUTE PERICARDITIS (Table 4)
Clinical features would depend upon the presence
or absence of effusion and upon the etiology of pericarditis. Features
most commonly encountered include chest pain, fever, and pericardial rub.
Chest pain. Pain is usually intense,
located anteriorly over the precordium or the retrosternal area. It is
described as pleuritic and is aggravated by deep breathing, lying supine,
or coughing. Pain most often radiates to the cervical area and the trapezius
ridge and less often to the abdomen and the interscapular area posteriorly.
The pleuritic pain causes the patients to breath in a shallow and rapid
manner i.e. tachypnea. It should not be confused with dyspnea, which
results from poor oxygenation, e.g. pulmonary congestion.
Physical examination. Detection of
pericardial
rub confirms the diagnosis of pericarditis. Pericardial rubs are more
commonly triphasic. The three components of the rub are a) atrial or presystolic
component, b) ventricular systolic component, c) ventricular diastolic
component; ventricular systolic component being the most prominent.
Electrocardiogram. Electrocardiogram
typically shows ST segment elevation in most leads followed, within days,
with return to baseline. This is then followed by T wave inversion and
finally within weeks, it returns to normal baseline. Occasionally PR segment
depression representing atrial injury may be noted.
Laboratory Tests & Other Tests.
Other tests are performed based on clinical suspicion of a specific etiology
or to exclude certain etiologies. These include LE, ANA (SLE), PPD skin
test (tuberculosis), chest x-ray, CT scan (malignancy), mammogram (breast
cancer), rheumatoid factor (rheumatoid arthritis). Serum viral titers are
obtained in acute phase and convalescent phase. An increase in titers is
highly suggestive of viral etiology. Pericardial effusion is tested for
cells, culture etc. as discussed later in this section.
TREATMENT OF PERICARDITIS
(Table 5)
The majority of cases of viral pericarditis and post-infarction
pericarditis resolve in 1-3 weeks. Analgesics i.e. non-steroidal agents
such as aspirin are used for relief of pain. Steroids are avoided and are
employed only in refractory cases. Other specific measures include drainage
of purulent pericardial effusion and antibiotics for bacterial pericarditis,
antituberculous therapy for tuberculous pericarditis, hemodialysis for
uremic pericarditis, drainage of effusion followed by local infiltration
of steroids in pericardial space for uremic pericarditis, radiation and
chemotherapy for pericarditis of malignancy, pericardiectomy for recurrent
pericarditis with effusion, tamponade due to malignancy, etc.
PERICARDIAL EFFUSION
It is common to note a small amount of effusion in
most patients with pericarditis. However, the amount of effusion may be
large under certain situations. Thus conditions that may lead to large
effusions include:
ETIOLOGY
1. Inflammation from infection, immunologic process.
2. Trauma causing bleeding in pericardial space.
3. Noninfectious conditions such as:
a. increase in pulmonary hydrostatic pressure e.g.
congestive heart failure.
b. increase in capillary permeability e.g. hypothyroidism
c. decrease in plasma oncotic pressure e.g. cirrhosis.
4. Decrease drainage of pericardial fluid due to obstruction
of thoracic duct as a result of malignancy or damage during surgery.
Effusion may be serous, serofibrinous, suppurative,
chylous, or hemorrhagic depending on the etiology. Viral effusions are
usually serous or serofibrinous; malignant effusions are usually hemorrhagic.
CLINICAL FEATURES
Although patients may be symptomatic from pericarditis,
a small amount of effusion does not produce hemodynamic abnormalities.
A large amount of effusion may lead to compression of adjoining structures
and produce symptoms of dysphagia, hoarseness, and hiccups. Compression
of lung leading to an area of consolidation (detected as percussion dullness
and bronchial breathing) in the left infrascapular region is known as -
Ewalt's sign.
CARDIAC TAMPONADE
The term cardiac tamponade is used to describe compression
of the heart by high intrapericardial pressure resulting from accumulation
of pericardial effusion. Factors that determine tamponade include 1) rate
of accumulation of pericardial fluid 2) amount of fluid 3) pericardial
compliance (Table 6).
A sudden increase in intrapericardial volume e.g.
100ml to 200ml of blood in pericardial space due, for example, to puncture
wound of the heart will cause a significant rise in the intrapericardial
pressure leading to tamponade. On the other hand a patient with tuberculous
pericarditis may accumulate as much as 1-2 liters of fluid before there
is evidence of tamponade. Similarly less fluid would be needed to cause
tamponade if the pericardium was thick and therefore noncompliant compared
to a compliant pericardium (refer to figure 1). Tamponade
is associated with decrease in cardiac filling, which leads to a decrease
in cardiac output. As a result of a decrease in cardiac output several
compensatory mechanisms are activated (see table 9),
notable amongst which is an increase in sympathetic activity. High circulatory
catecholamines serve several useful purposes, but at the expense of several
potentially deleterious effects which are indicated in parenthesis:
a) Venous tone is augmented which increases venous
pressure and facilitates cardiac filling. Increase in preload augments
stroke volume through the Frank-Starling mechanism. (High venous pressure
may lead to hepatomegaly, ascites, edema).
b) Arteriolar resistance is increased which helps
to support blood pressure.
(Increase in afterload increases myocardial work,
also decreases stroke volume).
c) Adrenergic mediated tachycardia helps to maintain
adequate cardiac output in the face of low stroke volume (increase in myocardial
oxygen consumption (MVO2).
d) Adrenergic mediated increase in contractility
improves stroke volume and ejection fraction (increase in MVO2).
With further increase in intrapericardial pressure,
these compensatory mechanisms fail resulting in severe hypotension and
subsequently to shock.
Hemodynamics of Tamponade (Table
8)
As discussed earlier, there is augmentation of blood
flow to the right ventricle during inspiration which results in two important
hemodynamic changes i.e. a decline in jugular venous pressure and a drop
in systolic blood pressure with inspiration (relative to expiration) i.e.
pulsus paradoxus (normal < 10mmHg).
In cardiac tamponade, although intrapericardial pressure
(IPP) is elevated, negative thoracic pressure is transmitted to fluid-filled
pericardial space and therefore, is a directional drop of IPP during inspiration
e.g. IPP of 25mmHg drops during inspiration to 10mmHg (producing a pulsus
paradoxus). It is exaggerated i.e. > 10mmHg by the combined effects
of the septal bulge and by the effect of high IPP on the left ventricle.
Thus the increase in RV (and decrease in LV) volume is maintained (and
exaggerated) during inspiration.
CLINICAL FEATURES OF TAMPONADE (Table
7)
Clinical features may vary depending on the stage
of tamponade. In advanced stages of tamponade clinical features include
hypotension, tachycardia, distended jugular veins and a pulsus paradoxus
of > 10mmhg. With progressive increase in intrapericardial pressure, there
is further elevation of venous pressure and a drop in cardiac output, which,
if untreated, leads to cardiogenic shock.
DIAGNOSIS
Pericardial effusion (and tamponade) should be suspected
in any patient with above clinical features who have had a prior history
of pericarditis or who have an illness that potentially leads to pericarditis
e.g. breast cancer, tuberculosis, hemodialysis, patients with AIDS. The
most useful test is echocardiography. This test helps to quantify the amount
of effusion and in establishing the presence or absence of tamponade. Tamponade
is indicated by diastolic collapse of the right atrium and sometimes the
right ventricle and is indicative of high intrapericardial pressure.
ELECTROCARDIOGRAM (see Table
10) may show low voltage and electrical alternans.
ECHOCARDIOGRAM: See Table
10
TREATMENT (Table 11)
Definitive therapy for tamponade is drainage of the
effusion by pericardiocentesis or by surgery. Pericardial fluid obtained
by pericardiocentesis or surgical drainage may be analyzed looking for
specific diagnosis and the myocardial biopsy specimen is examined histologically.
Volume expansion and inotropic therapy are used as temporary emergency
measures while preparing for pericardial drainage. Diuretics are contraindicated
(why?).
CONSTRICTIVE PERICARDITIS
(Table C1)
The hallmark of constrictive pericarditis is a thickened
adherent pericardial sac that restricts filling of the heart. The condition
is important since early recognition is surgically treatable. In addition
the condition can be confused with liver cirrhosis and with restrictive
cardiomyopathy with which it bears a close clinical resemblance.
ETIOLOGY (Table C1)
Almost any condition that causes acute pericarditis
can potentially lead to constrictive pericarditis. Recent studies indicate
surgery as a common cause constrictive pericarditis although it occurs
in a small percentage of patients. Other causes include viral, neoplastic
diseases such as lymphoma, infectious diseases such as tuberculosis and
bacterial infectious, chronic renal failure, radiation therapy, and rheumatoid
arthritis.
PATHOLOGY (Table C2)
Pericardial effusion that develops in the course
of acute pericarditis resolves in most instances. In some, the resolution
is followed by thickening and fibrosis or calcification of the pericardium
over a period of weeks, months or even years. These changes also involve
adhesion of the two pericardial layers, and the pericardial space in most
cases becomes obliterated.
PATHOPHYSIOLOGY
Noncompliant pericardium interferes with diastolic
filling of all cardiac chambers. Systolic function is normal. As the ventricles
begin to relax and fill from end systole, filling in the early (1/3)
diastolic phase is normal. Continued filling expands the ventricles until
they abruptly encounters the rigid pericardium and filling stops in later
(2/3) part of diastole (table C5). This filling pattern
produces the typical dip-plateau on the ventricular pressure tracings.
In addition since all chambers are equally affected by the rigid pericardium,
all end diastolic pressures are elevated to the same level i.e. equalization
of diastolic pressures. Although systolic function is normal, decrease
in ventricular filling reduces cardiac output. Elevation of right heart
diastolic pressure raises systemic venous pressure, which leads to jugular
venous distention, hepatomegaly, and edema.
In constrictive pericarditis the increase in venous
return to the thoracic region during inspiration cannot be accommodated
by the right ventricle, which is surrounded by noncompliant pericardium.
As a result the extra volume of blood distends the neck veins i.e. Kussmaul's
sign (table C4). At the same time failure of the right
ventricle to expand, does not affect the interventricular septum and therefore
left ventricular volume is not decreased and pulsus paradoxus is therefore
absent.
CLINICAL FEATURES (Table C3)
Decrease in cardiac output leads to poor perfusion
of exercising muscles and causes fatigue and dyspnea and sometimes syncope.
Jugular veins are prominent, hepatomegaly, ascites and edema are noted.
Auscultation of the heart may reveal a loud diastolic sound - pericardial
"knock", produced by abrupt cessation of ventricular filling by the stiff
pericardium. Kussmaul's sign is present and pulsus paradoxus is unknown.
DIAGNOSIS
Echocardiogram is useful in providing key hemodynamic
findings. The presence of pericardial effusion indicates effusive constrictive
pericarditis and may reveal thick pericardium. Electrocardiogram is abnormal
but nonspecific atrial fibrillation is common. Chest x-ray may show evidence
of pericardial calcification. CT and MRI are most helpful in differentiating
constrictive pericarditis from restrictive cardiomyopathy with which it
resembles so closely both hemodynamically and clinically. Cardiac catheterization
shows the typical dip-plateau pattern and equalization of diastolic pressures.
The difference between left atrial mean and right atrial mean pressure,
if present, amounts to less than 5mmHg.
TREATMENT
The most definitive therapy consists of pericardiotomy.
Results are best when the procedure is done in the early stage of the disease
and hence the importance of early recognition. Supportive measures include
diuretics, high protein diet, etc. Specific therapy is guided by diagnosis
e.g. tuberculosis.
COMPARISON OF HEMODYNAMIC FINDINGS OF
TAMPONADE VERSUS CONSTRICTION
Several subtle clinical and hemodynamic
differences can help to distinguish between cardiac tamponade and pericardial
constriction. These findings are summarized in table C6,
and include several features already described above.
Characteristic jugular venous pressure
tracings can be seen in these two syndromes which can also be helpful in
distinguishing between the two:
In cardiac tamponade, right ventricular
filling is impaired throughout diastole, and therefore the
y-descent (which represents the fall in right atrial pressure immediately
after tricuspid valve opening as blood rushes into the right ventricle)
is blunted (less steeply downsloping).
In pericardial constriction, the earliest
phase of diastolic right ventricular filling is not impaired and the y-descent
is not blunted -- in fact, because it is descending from a higher-than-normal
right atrial pressure, the y-descent may be accentuated, commonly
called a "rapid y-descent".
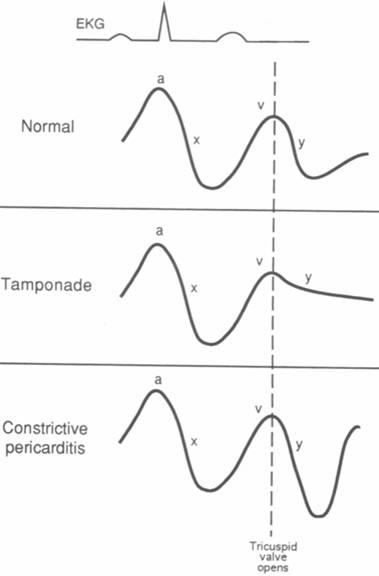
Schematic diagrams of right atrial
(or jugular venous) pressure recordings.
A. Normal. The initial a wave
represents atrial contraction. The v wave reflects passive filling of
the atria during systole, when the tricuspid and mitral valves are closed.
After the tricuspid valve opens, the right atrial pressure falls (y descent)
as blood empties into the right ventricle.
B. Cardiac tamponade. High-pressure
pericardial fluid compresses the heart, impairing right ventricular filling,
so that the y descent is blunted.
C. Constrictive pericarditis. The
earliest phase of diastolic filling is not impaired so that the y descent
is not blunted. The y descent appears accentuated because it descends from
a higher than normal right atrial pressure. The right atrial c wave is
not shown.
COMPARISON OF HEMODYNAMIC FINDINGS OF RESTRICTIVE
CARDIOMYOPATHY VERSUS CONSTRICTIVE PERICARDITIS:
The hemodynamic findings of restrictive
cardiomyopathy and those of constrictive pericarditis may be very
similar. Often, clinical characteristics are not enough to differentiate
between the two diagnoses, and information from echocardiography, CT scanning,
nuclear magnetic resonance imaging (MRI), or hemodynamic findings from
right and left heart catheterization must be employed. Characteristic findings
seen during cardiac catheterization are summarized in table
C7, below.
Many of these hemodynamic findings can
be explained by the fact that, in restrictive cardiomyopathy there tends
to be systolic impairment of both ventricles, while in constrictive pericarditis
there is normal left ventricular systolic muscle function -- hence there
tends to be normal PCW pressure. In addition, with constriction there
tends to be more equalization of diastolic pressures due to the rigid "shell'
around the heart -- and hence the left atrial (i.e. PCW) pressure
is almost identical to the right atrial pressure. Conversely, in restriction
there is usually a small difference in RA and LA pressures, which
can be accentuated by increasing preload.
NORMAL ANATOMY OF THE PERICARDIUM
1. INNER VISCERAL LAYER
2. OUTER PARIETAL LAYER
3. PERICARDIAL SPACE
4. PERICARDIAL FLUID
FUNCTIONS OF THE PERICARDIUM
ANCHORING
PROTECTION
PREVENT EXCESSIVE DILATION
PATHOLOGY OF ACUTE PERICARDITIS
FIBRIN DEPOSITION
("BREAD & BUTTER" APPEARANCE)
LEUKOCYTE PRODUCTION
PERICARDIAL EFFUSION
CLINICAL FEATURES OF ACUTE PERICARDITIS
-
CHEST PAIN
-
FEVER +
-
TACHYPNEA
-
PERICARDIAL RUB
-
ELECTROCARDIOGRAM
-
ECHOCARDIOGRAM
-
LABORATORY STUDIES
TREATMENT OF ACUTE PERICARDITIS
· NONSTEROIDALS
· STEROIDALS
· SPECIFIC
· D/C SUSPECT DRUGS
Cardiac Tamponade
FACTORS CONTRIBUTING TO CARDIAC
TAMPONADE
RATE OF ACCUMULATION
SIZE OF EFFUSION
PERICARDIAL DISTENSIBILITY
VENTRICULAR DIASTOLIC COMPLIANCE
HEMODYNAMICS OF PERICARDIAL
EFFUSION
RELATIONSHIP OF CHRONICITY
TO HEMODYNAMICS
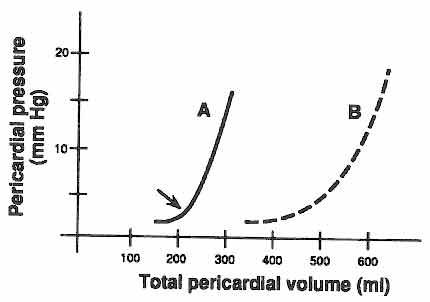
A - Acute Effusion, e.g. traumatic injury of the
heart
B - Chronic Effusion, e.g., viral pericarditis, tuberculosis
The normal pericardium is rigid but does possess
the capacity to stretch in response to increase in pericardial effusion.
However this ability to stretch is influenced by time and volume. In the
example above, if the pericardial volume is increased suddenly (Graph A),
the ability to stretch is limited and the IPP rises. On the other hand
if the volume is increased gradually, the pericardium has time to stretch
and the IPP does not rise until a large volume has accumulated in the pericardial
space (Graph B).
CLINICAL FEATURES OF CARDIAC
TAMPONADE
-
VARIABLE
-
WEAKNESS
-
DIAPHORESIS
-
TACHYPNEA
-
DYSPNEA
-
NEAR SYNCOPE
-
JVP
-
KUSSMAUL SIGN ABSENT
-
PULSUS PARADOXUS (>10mmHg)
HEMODYNAMICS OF CARDIAC COMPRESSION
|
DECREASED LV FILLING
|
DECREASED RV FILLING
|
|
¯¯
|
¯¯
|
|
DECREASED LV STROKE VOLUME &
CO
|
DECREASED RV STROKE VOLUME
& CO
|
|
¯¯
|
¯¯
|
|
HYPOTENSION
|
VENOUS PRESSURE
|
|
¯¯
|
¯¯
|
|
¯
PERIPHERAL PERFUSION
|
JUGULAR VENOUS DISTENTION
|
|
¯¯
|
¯¯
|
|
FATIGUE, DYSPNEA, TACHYCARDIA,
MENTAL OBTUNDATION, OLIGURIA
|
HEPATOMEGALY, ASCITES, EDEMA
|
N.B.:
NO PULMONARY CONGESTION
DESPITE
PULM VENOUS PRESSURE
COMPENSATORY CHANGES IN CARDIAC
TAMPONADE
INCREASED CATECHOLAMINES:
BENEFIT:
CONTRACTILITY ®
EF
HR ®
CO
VENOUS
TONE ®
PRELOAD
®
SV
PERIPH
ART RESIST ®
BP
HARM:
MVO2
AFTERLOAD
ARRHYTHMIAS
ACTIVATION OF RENIN ANGIOTENSIN ALDOSTERONE SYSTEM:
BENEFIT: Na + H20
®
PRELOAD
® SV
ANGIO II ®
ART RESISTANCE ® BP
HARM: EDEMA
AFTERLOAD
ELECTROCARDIOGRAPHIC FINDINGS IN TAMPONADE
ELECTRICAL ALTERNANS
LOW VOLTAGE
ECHOCARDIOGRAPHIC FINDINGS IN TAMPONADE
EVIDENCE OF EFFUSION
RA, RV DIASTOLIC COLLAPSE
"SWINGING" OF HEART
TREATMENT OF CARDIAC TAMPONADE
GENERAL
VOLUME EXPANSION
INOTROPIC SUPPORT
AVOID DIURETICS
DEFINITIVE
PERICARDIAL FLUID DRAINAGE
PERICARDIOCENTESIS
PERICARDIAL "WINDOW"
SURGICAL RESECTION
Constrictive Pericarditis
-
CONSTRICTION OF THE HEART BY THICKENED, FIBROSED
OR CALCIFIC PERICARDIUM
-
RESTRICTION TO FILLING OF THE HEART
The hallmark of CP is a thickened, fibrosed and often
calcific pericardium that is adherent to the epicardial surface and causes
a variable degree of cardiac compression. There is impediment to ventricular
filling due to the restraining effect of the noncompliant pericardium.
Systolic function is normal. Therefore CP is a form of diastolic heart
failure (as is also restrictive cardiomyopathy).
ETIOLOGIES OF CONSTRICTIVE PERICARDITIS
INFECTIOUS:
POST-VIRAL PERICARDITIS
POST-BACTERIAL PERICARDITIS
TUBERCULOSIS
FUNGUS (HISTOPLASMOSIS)
INFLAMMATORY:
POST-MI (DRESSLER SYNDROME)
POST-PERICARDIOTOMY SYNDROME
POST-TRAUMA
RHEUMATOID ARTHRITIS
COLLAGEN VASCULAR:
SYSTEMIC LUPUS ERYTHEMATOSUS
MALIGNANT:
METASTATIC MALIGNANCY
MISCELLANEOUS:
POST-RADIATION
UREMIA
PATHOLOGY OF CONSTRICTIVE
PERICARDITIS
FIBROSIS
CALCIFICATION
ADHESION
SIGNS AND SYMPTOMS OF CONSTRICTIVE
PERICARDITIS
1. ELEVATED VENOUS PRESSURE:
JUGULAR VENOUS DISTENTION
HEPATOMEGALY
ASCITES
EDEMA
2. LOW CARDIAC OUTPUT
DYSPNEA
FATIGUE
TACHYCARDIA
3. CARDIAC EXAM:
DISTANT HEART SOUNDS
PERICARDIAL "KNOCK"
CHARACTERISTIC HEMODYNAMIC
CHANGES WITH RESPIRATION
INSPIRATION
VENOUS RETURN TO THORAX
FAILURE OF RV FILLING TO
(Due to non-compliant pericardium)
JVP RISES (i.e. +
Kussmaul's Sign )
N.B.: PULSUS PARADOXUS ABSENT
CHANGES DURING CARDIAC
CYCLE WITH CONSTRICTIVE PERICARDITIS
HEMODYNAMICS
SYSTOLE
NORMAL SYSTOLIC
FUNCTION
DIASTOLE
FIRST PART: NORMAL FILLING
LATER PART: FILLING
STOPPED BY THICK PERICARDIUM
RESULT:
DIP & PLATEAU
PRESSURE TRACING ("SQUARE ROOT SIGN")
CARDIAC COMPRESSION:
CLINICAL COMPARISON OF TAMPONADE VERSUS CONSTRICTION
|
Tamponade |
Constriction |
| Pericardial calcification |
Absent |
Common |
| Kussmauls sign |
Absent |
Common |
| Jugular pulse tracing |
Xy or XY |
XY or xY |
| Paradoxic pulse (>10mmHg) |
Present |
Rare |
| Abnormal S3
("knock") |
Absent |
Common |
| Cardiac Cath Square Root
Sign |
Absent |
Present |
| Atrial Fibrillation |
Rare |
Common |
HEMODYNAMIC DIFFERENCES
BETWEEN RESTRICTIVE CARDIOMYOPATHY
AND CHRONIC CONSTRICTIVE
PERICARDITIS
| |
RESTRICTIVE CARDIOMYOPATHY
|
CONSTRICTIVE PERICARDITIS
|
|
CARDIAC OUTPUT
|
Usually Decreased
|
Normal or Decreased
|
|
PCW
|
Increased
|
Normal
|
LA - RA
(DIFFERENCE IN MEAN LEFT AND RIGHT ATRIAL
PRESSURES)
|
> 6mmHg
|
< 6mmHg
|
|
RV DIASTOLIC PRESSURE
|
Diastolic Plateau elevated to
>
1/3 systolic pressure
|
Diastolic plateau rarely 1/3
systolic pressure
|
PA SYSTOLIC / RA
(RATIO OF SYSTOLIC PULMONARY ARTERY PRESSURE
TO MEAN RIGHT ATRIAL PRESSURE)
|
> 3.5 : 1
|
< 3.5 : 1
|
|
RESPIRATORY VARIATION IN PRESSURES
|
Usually present
|
Often absent
|
|
Change in LVEDP (or PCW) with exercise
|
Rises markedly
|
Minimal rise
|
Legend
PCW = PULMONARY CAPILLARY WEDGE PRESSURE;
LA = Left atrium; RA = Right Atrium; PA = Pulmonary Artery

