News
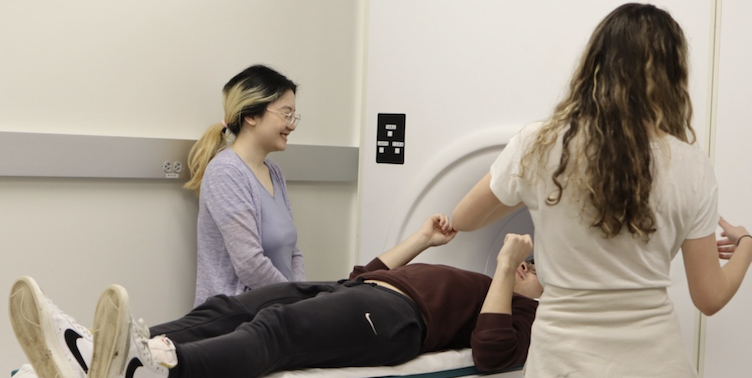
‘ScanFest’ sessions teach students the ins and outs of using an MRI machine for research
As part of a class taught by Brown neuroscientist David Badre, undergraduates embrace the rare op

Carney Institute grants more than $600,000 in innovation awards to Brown University scientists
This year’s projects span behavioral and systems neuroscience, circuit therapeutics

Carney researchers show that large language model AIs can reproduce mechanisms found in the human brain
A collaboration between professors Michael Frank and Ellie Pavlick is yielding impo

Brown researchers develop brain-inspired wireless system to gather data from salt-sized sensors
The novel approach helps advance wireless sensor technology and paves the way for one day using l

Virtual reality exposure plus electric brain stimulation offers a promising treatment for PTSD
Results from a clinical trial show that an innovative combination of two treatments can be an eff

Marvel at the mysteries of the human brain at the Brown Brain Fair on March 16
Free and open to the public, the annual family-friendly event invites Rhode Islanders to learn ab
Acting on Impulse: Parkinson’s disease treatments alter decision-making through two paths
Dopaminergic therapy and deep brain stimulation can improve the motor symptoms of Parki
A semester-long program at ICERM and a dynamic new hire at Carney promise a major payoff for brain science
Mathematical neuroscientist Carina Curto led an ambitious semester program at ICERM

Elon Musk’s Neuralink Has Implanted Its First Chip in a Human Brain. What’s Next?
"The thing is, you’ve got to be careful.
