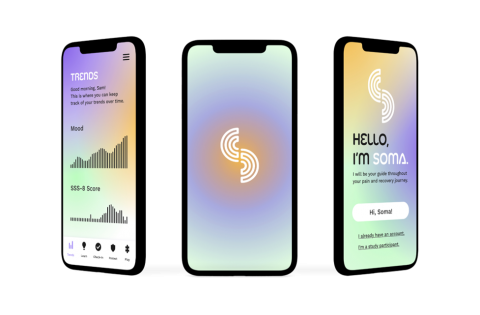
Chronic pain is the unseen giant among health afflictions, quietly eclipsing the impact of heart disease, cancer, and diabetes combined. In America, this translates to a staggering one in five people living with this relentless condition. This rise signals an urgent call to action, as the current prospects for those suffering are far from promising: While traditional pharmacological treatment approaches are adequate for acute pain, they fall short when it comes to chronic conditions—often leading to much more invasive procedures. Regrettably, even the most aggressive treatments, such as spinal cord surgeries or radiofrequency ablation, fail to alleviate the suffering for over half of the patients treated, resulting in countless individuals enduring pain for years, or even decades, without suitable relief. Why do most treatments for Chronic Pain fail? The answer may well be within the brain. Emerging neuroscience research suggests that chronic pain is fundamentally different from acute pain, stemming from alterations in brain circuits that govern learning. This suggests that for many, chronic pain could be less an unhealed injury and more a learned persistent pattern. This insight reveals why conventional treatments, which target the periphery, but not the brain, are so often inefficient. Effective approaches, like cognitive and behavioural therapies, can alter the brain's response to pain, but access to such treatments is scarce, leaving many without the appropriate care.
Recognizing this gap, our interdisciplinary team at Brown University—comprising neuroscientists, physicians, and software engineers—has spent the last two years developing SOMA. SOMA represents a cutting-edge fusion of user-centric technology and neurobiological research designed to dismantle the barriers of chronic pain management. Through an intuitive smartphone app, SOMA harnesses user-input data on symptoms and medication, alongside cognitive assessments and mind-body exercises that are currently being developed into an engaging and informative digital therapeutic. The data are used in mechanistic computational models and machine learning algorithms to predict individual pain trajectories and treatment outcomes, in order to inform users about their pain trends, develop digital phenotypes and biomarkers and tailor interventions to an individual's needs. The first version of SOMA recently released to the App stores has already garnered over 1500 users, many of whom have battled chronic pain for over a decade. Our project has received several prizes and grants and was featured in the news. Through SOMA we aspire to empower patients and to transform chronic pain care into a personalized, efficacious, and universally accessible service, providing long-sought relief for countless individuals.
Get in Touch
Interested in staying up to date? Sign up for our Mailing List
Want to download the App? Here is a link to the AppStore and GooglePlay Store
Team
Prof. Dr. Frederike Petzschner
Chloe Zimmerman
Bradfoard Roarr
Find the full team here
Recent Updates on SOMA:
February 2021: Work on SOMA begins
October 2021: SOMA get accepted into the NEMIC Med Tech Leadership Program
May 2022: SOMA win’s the Pitch Competitions of the Med Tech Leadership Program (Award: 500 USD) Here are pictures of the event and the announcement from NEMIC
June 2022: SOMA gets accepted into the NEMIC Activate Program (Award: 15 000 USD)
August: 2022: A Science Project around SOMA receives funding from the NIH COBRE (Funding: 250 000 USD)
March 2023: A paper on SOMA is published as a PrePrint
March 2023: SOMA is released on the Google Play and Apple App Store (Link: AppStore and GooglePlayStore)
April 2023: The first external clinical study was launched on SOMA
April 2023: Press Release
May 2023: Boston Globe covers SOMA
August 2023: Funding from the COBRE was renewed (Funding: 200 000 USD)
August 2023: SOMA receives the BBII Award (Funding: 100 000 USD)
September 2023: SOMA get accepted into the MIT Faculty Founder Program (Award: 10 000 USD)
October 2023: SOMA reaches over 1500 downloads for the App Stores