Decision Chart | Comprehensive Comparison Table | Definitions | Public Health Surveillance
Brown's IRB has assured federal regulatory agencies that the institution will review and approve all research that meets the federal definition of human subject research. Determining whether or not a project meets the federal definition of human subjects research is a two-step process. The investigator must first determine if the project meets the federal definition of research and, if so, then determine if the project includes human subjects.
In addition to the information below, the Brown HRPP has provided self-assessment resources for students and researchers to use to determine if their project requires IRB review:
- The Self-determination tools and checklists are guides to assist Brown researchers in complying with federal regulations and institutional policy.
- The NIH Decision Tool: Am I Doing Human Subjects Research? Developed by NIH to help you determine if your research involves human subjects, may be exempt from federal regulations, or is not considered human subjects research.
-
Is it Human Subjects Research? Is IRB Review Required? educational video from HRPP's Learning Library.
Decision Chart
A decision chart to help guide researchers in making a determination of whether they're conducting human subject research and need to submit an IRB Application.
Step 1: Is your Project Considered Research?
Federal regulation defines "research" as a systematic investigation, including research development, testing, and evaluation, that is designed to develop or contribute to generalizable knowledge.
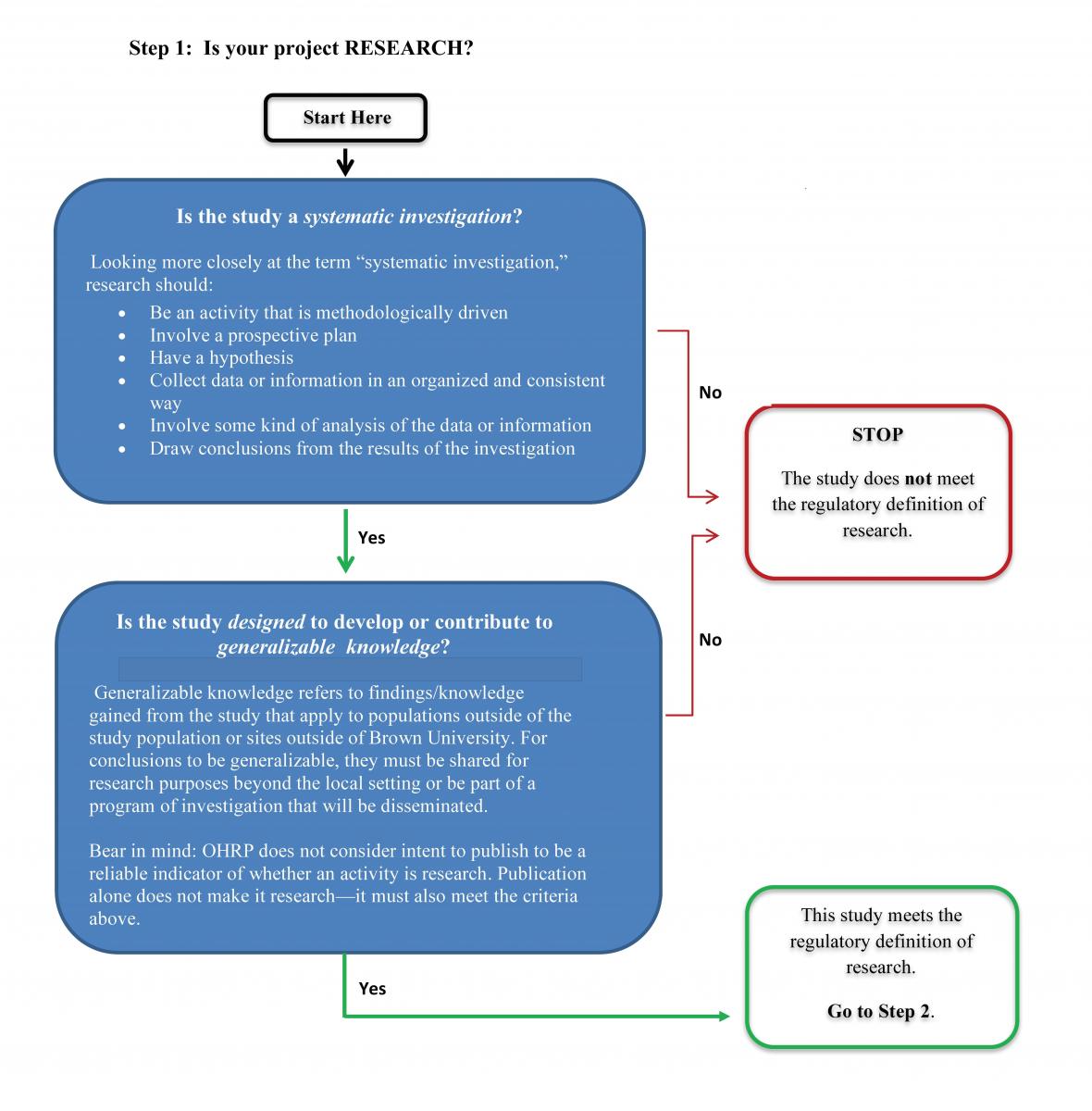
Systematic Investigation
A "systematic investigation" is a detailed or careful examination that has or involves a prospectively identified approach to studying a specific topic, answering a specific question(s), testing a specific hypothesis(es), or developing theory based on a system, method, or plan. Systematic investigations include observational studies, interview or survey studies, group comparison studies, test development, and interventional research. Projects that are not systematic investigations include, for example, oral histories, journalism, and phenomenological activities. Program evaluation is seen as a gray area and requires further assessment of design and intent.
Generalizable knowledge
Developing or contributing to "generalizable knowledge" means that the intent or purpose of the systematic investigation is to produce knowledge from which conclusions will be drawn that can be applied to populations outside of the specific study population. This usually includes one or more of the following concepts:
- knowledge that contributes to a theoretical framework of an established body of knowledge;
- the primary beneficiaries of the research are other researchers, scholars, and practitioners in the field of study;
- dissemination of the results is intended to inform the field of study (this alone does not make an activity constitute research “designed to contribute to generalizable knowledge”);
- the results are expected to be generalized to a larger population beyond the site of data collection;
- the results are intended to be replicated in other settings.
If your project is not considered research, you do not need to submit an IRB application. If your project does meet the definition of research, proceed to Step 2.
_______________________________________________________________________________________________________________________________________________________
Step 2: Does it involve human subjects?
The Federal Policy for the Protection of Human Subjects (Common Rule) defines a human subject as “…a living individual about whom an investigator (whether professional or student) conducting research obtains (1) data through intervention or interaction with the individual, or (2) identifiable private information.”
Examples that do not meet the definition of Human Subjects:
- Analysis of data about people who are deceased
- Secondary analysis of anonymous data
- Interviews with “key informants” about topics other than themselves
Note that the definition of human subject focuses on what information is obtained about people or material that is acquired from people. If either of the following is true, your research activity involves human subjects:
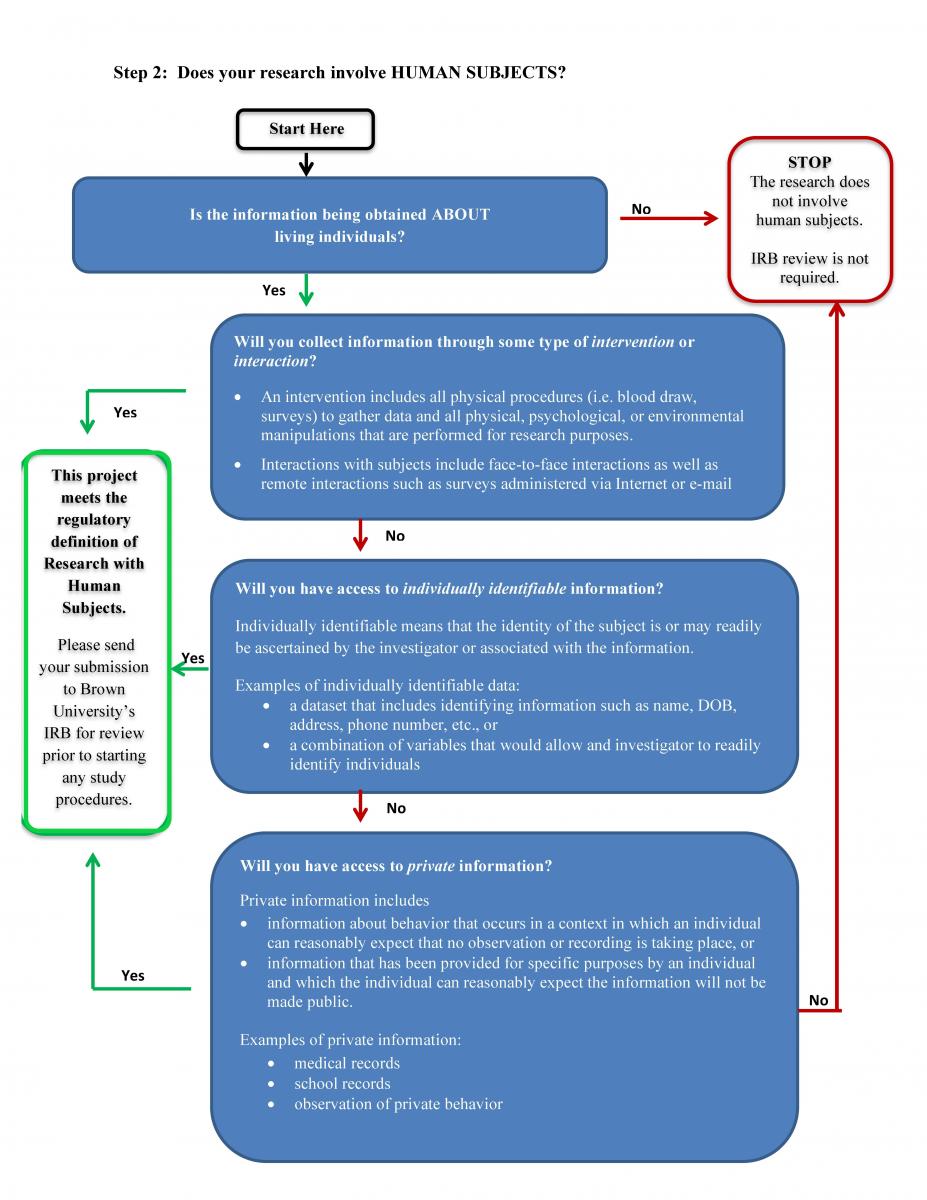
If your project does not include human subjects, you do not need to submit an IRB application.
Comprehensive Comparison Table
A comprehensive comparison table to assist students and researchers with distinguishing whether a project qualifies as human subject research, quality assurance/quality improvement, program evaluation, a student project or scholarly & journalistic activities.
HUMAN SUBJECT RESEARCH
|
QUALITY IMPROVEMENT OR ASSESSMENT
|
PROGRAM EVALUATION
|
STUDENT PROJECT
|
SCHOLARLY & JOURNALISTIC ACTIVITIES
|
|
|---|---|---|---|---|---|
INTENT
|
Common Rule: “Research means a systematic investigation, including research development, testing and evaluation, designed to develop or contribute to generalizable knowledge.”
Belmont Report: “[T]he term ‘research’ designates an activity designed to test a hypothesis, permit conclusions to be drawn, and thereby to develop or contribute to generalizable knowledge (expressed, for example, in theories, principles, and statements of relationships). Research is usually described in a formal protocol that sets forth an objective and a set of procedures designed to reach that objective.”
OHRP: “The question "what is research" frequently arises in relation to an investigator or institutional activity being planned to gather data to evaluate a specific program, such as a QA/QI activity. Although the determination as to whether the activity will contribute to 'generalizable knowledge' is often based on whether the data will be disseminated by means of publication or presentation, this should not be the sole factor used to make the determination. In general, OHRP gives guidance that if the data will be used to draw conclusions related to a larger entity, then the activity is considered 'research'.” |
Intent of the project is to improve a practice or process within a particular institution or ensure it conforms to expected norms. |
Intent of the project is to evaluate a specific program, only to provide information for and about that program. |
Intent of the project is to provide an educational experience about the research process or methods |
This category of activities concerns certain activities in various fields that focus directly on the specific individuals about whom information are collected (e.g., oral history, journalism, biography, literary criticism, legal research, and historical scholarship).
Literary criticism …because while a piece of literary criticism might focus on information about the author(s), it would typically focus on the specific author(s) in view.
Legal research… because it would often focus on the circumstances of specific plaintiffs or parties involved in a case.
It is not the particular field that removes the activity from the definition [of HSR], but rather the particular activity's focus on specific individuals. |
MOTIVATION FOR PROJECT
|
Project occurs in large part as a result of individual professional goals and requirements (e.g., seeking tenure; obtaining grants; completing a thesis or dissertation). |
Project occurs regardless of whether individual(s) conducting it may benefit professionally from conducting the project. |
Project not initiated by the evaluator and occurs regardless of whether individual(s) conducting it may benefit professionally from conducting the project. |
Project occurs as part of assigned course/class work or a requirement of an educational program in order to learn a new technique or pass a course/fulfill an assignment. |
Project occurs to portray the individuals involved. |
DESIGN
|
Designed to develop or contribute to the scientific storehouse of knowledge; may involve randomization of individuals to different treatments, regimens, or processes; novel research ideas or experimental activities that are not yet known to be efficacious. May be designed to prove a relationship or correlation. |
Not designed to develop or contribute to generalizable knowledge; generally does not involve randomization to different practices or processes. |
Not designed to develop or contribute to generalizable knowledge; does not involve randomization of individuals, but may involve comparison of variations in programs |
Not designed to develop or contribute to generalizable knowledge; design is often an example or template provided by a professor or course book. |
Designed to collect and use information about specific individuals themselves, and not generalizing to other individuals. |
MANDATE
|
Activities not mandated by institution or program. |
Activity mandated by the institution or clinic as part of its operations. |
Activity mandated by the program, usually its funder, as part of its operations. |
Activity mandated by regularly assigned coursework or educational program |
Activities not necessarily mandated by institution or program |
EFFECT ON PROGRAM OR PRACTICE EVALUATED
|
Findings of the study are not expected to directly or immediately affect institutional or programmatic practice, although they may also be used for this purpose. Activity will be used to develop a problem statement, research questions, and/or theory-based hypotheses. |
Findings of the study are expected to directly affect institutional practice and identify corrective action(s) needed. |
Findings of the evaluation are expected to directly affect the conduct of the program and identify improvements. |
Findings of project are not expected to directly affect the program; the project will mainly generate raw data, not generalizable knowledge. |
N/A |
SUBJECT POPULATION
|
Human subject means a living individual about whom an investigator (whether professional or student) conducting research obtains (1) Data through intervention or interaction with the individual, or (2) Identifiable private information. May involve a subset of individuals; universal participation of an entire population (e.g., clinic, program, or department) is uncommon; generally, statistical justification for sample size is used to ensure endpoints can be met. |
Information on all or most receiving a particular treatment or undergoing a particular practice or process expected to be included; exclusion of information from some individuals significantly affects conclusions |
Information on participants receiving a particular treatment or undergoing a particular practice or process expected to be used; exclusion of information from some individuals significantly affects conclusions |
Can either include all, most, or a subset of individuals; statistical justification may be used in the context to understand the process of subject selection; however, recruitment often utilizes convenience sampling. |
Preamble to revised Common Rule: “… the focus is on the specific activities that collect and use information about specific individuals themselves, and not generalizing to other individuals, and that such activities occur in various fields of inquiry and methodological traditions… It is not the particular field that removes the activity from the definition, but rather the particular activity's focus on specific individuals.” |
BENEFITS
|
Participants may or may not benefit directly – benefit, if any, to individuals is likely to be incidental or delayed. |
Participants expected to benefit directly from the activities. |
No benefit to participants expected; evaluation concentrates on program improvements or whether the program should continue. |
Participants may or may not benefit directly; benefit is primarily for the student conducting the project for the fulfillment of educational requirements. |
Individual benefit may or may not be anticipated. |
DISSEMINATION OF RESULTS
|
Intent to publish or present generally presumed at the outset of project as part of professional expectations, obligations; dissemination of information usually occurs in research/scientific publications, grant proposals, or other research/scientific forum; results expected to develop or contribute to generalizable knowledge by filling a gap in scientific knowledge or supporting, refining, or refuting results from other research studies. Results of the project will be disseminated outside the institution for the purpose of sharing the outcomes or implications of the project, not just the process. |
Intent to publish or present generally not presumed at the outset of the project; dissemination of information often does not occur beyond the institution evaluated; dissemination of information may occur in quality improvement publications; when published or presented to a wider audience, the intent is to suggest potentially effective models, strategies, assessment tools or provide benchmarks or base rates rather than to develop or contribute to generalizable knowledge. |
Intent to publish or present generally presumed at the outset of the project; dissemination of information to program stakeholders and participants; may be publicly posted (e.g., website) to ensure transparency of results; when published or presented to a wider audience, the intent is to suggest potentially effective models, strategies, assessment tools or provide benchmarks or base rates rather than to develop or contribute to generalizable knowledge. |
Any presentations, posters, or publishing (including online) is simply to document the educational experience or completion of programmatic requirements. |
Intent to publish or present generally presumed at the outset of the project; dissemination of information occurs to document a specific historical event or the experiences of individuals without intent to draw conclusions or generalize findings. |
Definitions
Data about living individuals through intervention or interaction
An intervention may be physical procedures (e.g. venipuncture) or manipulations of living individuals or the living individuals’ environments.
An interaction may be communication or interpersonal contact between the investigator (or research team) and the living individual. Examples include interviews, questionnaires, surveys, observations, manipulations of subject behavior, diet, or environment, physical measurements, specimen collection (e.g. blood tissue), and administration of experimental drugs or devices.
Why “about whom” is key
Consider if the project focuses on the person or if the focus is on policies, practices or procedures about which the person is knowledgeable. Projects which collect information about policies, practices or procedures – even if the person who provided that information is identified – do not constitute human subject research. Asking a person about someone else does not make that person a human subject.
Identifiable private information about living individuals
Identifiable means 1) the identity of the individual from whom the information was obtained is ascertained or may be readily ascertained by the investigator; or 2) the identity of the individual from whom the information was obtained is associated or may be readily associated with the information.
Private Information is information about behavior that occurs in a context in which the individual can reasonably expect that no observation or recording is taking place or information that has been provided for specific purposes that the individual can reasonably expect will not be made public (e.g. medical record, employee or student records).
Examples of identifiable, private information include the subject’s name, address, phone number, social security number, medical record number, student or employee identification number, or in some cases, the combination of data such that they can identify a single individual through deductive reasoning. For example, data about employer, job title, age and gender may not individually identify a subject, but when combined, could in certain cases, identify a specific individual.
What is NOT considered identifiable, private information: If the information cannot be linked to a living individual, or is considered public or is given with the expectation that it will be made public and that it will be linked to the individual (e.g. biography or news story), then it would not be considered private identifiable information. For example, use of a publicly available data set that does not contain identifiers or codes linked to individuals does not involve human subjects research. However, use of a publicly available data set that does contain identifiers or codes linked to individuals does involve human subject research.
If you obtain/purchase/are given specimens/cells/material/data that has already been collected by someone else for some other purpose, and the specimens/cells/material/data are not linked to any identifiers that would make it reasonably possible to identify an individual, the activity is not considered research with human subjects.
If your activity does not involve human subjects as defined in the regulations, your activity does not fall under the purview of the IRB. You do not need to submit an application.
If you have determined that your research does meet the federal definition for human subjects research, you will need to apply for IRB review and approval before you begin (the IRB can not review projects retrospectively).
Still unsure or require documentation that your project does not need IRB review?
If you are unsure if your project meets the definition of research, or if you require documentation that your project does not require IRB review, please contact the HRPP at (401) 863-3050 or [email protected] to discuss. If you are a student, we strongly encourage you to first work with your advisor / mentor to discuss whether the proposed project meets the definition of human subjects research before contacting the HRPP.
Questions?
If you have questions, please contact us at 401-863-3050 or [email protected].
