



|

|
|
Dept. of Ecology and Evolutionary Biology
Brown University
80 Waterman Street, Box G-W
Providence, RI 02912

Lab: (401) 867-1032
Cell: (612) 386-0042
Curriculum Vitae (PDF,88KB)
|


|

|

|
|

2006-Present: Graduate student
Ecology and Evolutionary Biology, Brown University
2002-2006: B.S., Fisheries Science, Biology minor
Fisheries, Wildlife, and Conservation Biology, University of Minnesota

I am broadly interested in the evolution and morphology of fishes. This includes everything form the biomechanics of feeding and locomotion to molecular systematics. More specifically, I am interested in using phylogenetic trees to examine how key adaptations (such as the pharyngeal jaw apparatus, see below) can be specialized for different functions (such as molluscivory versus detritivory).
|

|

|
|
Biomechanics of the pharyngeal jaw apparatus
Prey capture and processing has long been of interest to all biologists, biomechanists and ecologists alike. Indeed, it is fundamental to the survival of any organism. Many vertebrates use only an oral set of jaws to capture and process their prey. Hyenas, for example, attack, subdue, and masticate their food in this way using oral jaws. Many fishes, in contrast, have two sets of jaws that are used in feeding. This second set of jaws, termed pharyngeal jaws (as they are located in the pharynx), is made up of modified gill arches that are hyper-ossified and have tooth-like structures. This second set of jaws allows for greater specialization, as the oral jaws (the only kind possessed by hyenas) can be specialized for prey capture and the pharyngeal jaws for prey mastication/processing.
Pharyngeal jaws have been variously modified in different groups of fishes. In the family Cyprinidae (commonly known as minnows and carps), for example, the entire fifth gill arch has been ossified into a single bone, from which only a few teeth protrude (see photo of the Cyprinid Opsopoeodus eimiliae to the right). These teeth are thought to act against the base of the skull (on a part of the skull termed the basioccipital process) as well as interdigitating with the teeth from the other arch (on the opposite side of the pharynx). In most perciform (perch-like) fishes, the pharyngeal jaws are formed by the modification of two or more gill arches, while each arch can consist of multiple elements that articulate with each other.
Due to the fact that these jaws are located inside the pharynx of the skull, they are difficult to visualize in a live fish during feeding. I will be using a new technology called CTX (developed by a group of researchers here at Brown) to study these enigmatic structures. CTX involves the use of 2 x-ray video cameras in conjunction with CT scanning to digitally reconstruct skeletal elements in four dimensions (3-D plus time). I hope to study a variety of taxa in order to gain a better understanding of both how these jaws are functioning and how those functions have evolved.
|

|
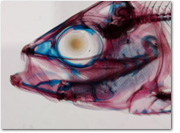
A cool fish head!
|
|
Underground burrowing by sand lances
Throughout the animal world, the ability to escape from potential predators is vital for survival. Sand Lances (family Ammodytidae) have evolved a curiously novel mechanism for escaping their predators. These normally pelagic (water column-dwelling) fish will swim rapidly downwards and continue swimming even when they hit the sandy substrate over which they normally occur. This swimming motion allows them to completely dive into sand, thereby escaping potential dangers. The exact way in which they dive into the sandy substrate has only been cursorily investigated. When I examined this fascinating behavior using high-speed video, only the above ground portions could be quantified. Click **HERE** for a slow-motion video of a Pacific Sand Lance, Ammodytes hexapterus, diving. Given that these burial events occur into sand, the mechanics for locomotion under the substrate have never been easily investigable. I will be using x-ray video fluoroscopy to examine subterranean locomotion in the Atlantic Sand Lance, Ammodytes americanus. This new technique will help in gaining a better understanding of the mechanics of anguilliform locomotion that these fish employ throughout this behavior. I will also compare the kinematics of underground locomotion with those of water column locomotion. Insight into the mechanics of burrowing and how they relate to our understanding of transitions in locomotor realm (i.e. swimming in water and in a sand-water mix) will allow for greater understanding in the burrowing of other vertebrates as well.
|

|
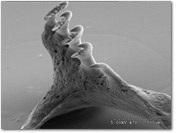
A Scanning Electron Microscope (SEM) picture of the pharyngeal arch in Opsopoeodus emiliae, the pugnose minnow. Note the serrations on the tooth cusps and the prominent, hooked tip of each cusp.
|
|
Cypriniform systematics and the evolution of herbivory in minnows
As an undergraduate, I had the opportunity to study in Dr. Andrew Simons' laboratory at the University of Minnesota. I worked as a research assistant on the Cypriniform Tree of Life grant as well as other molecular and morphological research projects. My undergraduate honors thesis was titled Character Suites Suggest Frequent, Punctuated Evolution of Herbivorous Morphologies in Minnows (Actinopterygii: Cyprinidae). This project involved using phylogenetic trees (one that we generated for this study and two that were generated in the context of different projects) and mapping morphological characters associated with diet onto those phylogenies. We found that shifts to an herbivorous diet were more common than previously supposed, and that the suite of character states exhibited by herbivorous fishes were independently derived several times in the minnow clade.
|

|
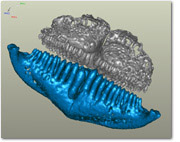
A CT scan of the upper (gray) and lower (blue) pharyngeal jaw elements of a convict cichlid, Archocentrus nigrofasciatus. Note that the upper and lower jaws interact with each other instead of the base of the skull.
|
|
Research skills and techniques
As an undergraduate, I gained experience with field collection and identification of freshwater fishes, molecular systematics, and various morphological techniques such as Low Viscosity nitrocellulose histology, Scanning Electron Microscopy, double staining & clearing, macro- and micro-dissection. As a graduate student I have been exposed to (and am still learning) sonomicrometry, high-speed video, X-ray imaging, and data analysis programs such as MatLab, Amira, and Maya.
|

|
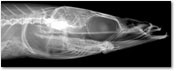
An X-ray still of the head of an American sand lance, Ammodytes hexapterus.
|
|
Teaching interests and experience
I've had the opportunity to TA two courses thus far. I taught general biology for two semesters at the University of Minnesota, after which I helped to develop a course called "Ecological Evaluation of Environmental Problems." I taught this course for two semesters as well. This course was especially rewarding for me, as I was able to bring students out into streams, lakes, and forests that were all within the metro area. Students were exposed to diversity that many of them would never have guessed existed. The course also involved discussions of environmental ethics, conservation, and methods for qualifying and quantifying biological diversity (as well as anthropogenic impacts on that diversity). I have found teaching to be very rewarding, as it gives me a chance to share my enthusiasm for biology (especially fish biology) with people of all backgrounds, some who may not have been previously interested in biology.
|

|

Teaching a section from "Ecological Evaluation of Environmental Problems" in Hiawatha creek, Minneapolis, MN
|
|


![]()
![]()







