PROVIDENCE, R.I. [Brown University] — A new study in the journal Nature shows how metals can be patterned at the nanoscale to be more resistant to fatigue, the slow accumulation of internal damage from repetitive strain.
The research focused on metal manufactured with nanotwins, tiny linear boundaries in a metal’s atomic lattice that have identical crystalline structures on either side. The study showed that nantowins help to stabilize defects associated with repetitive strain that arise at the atomic level and limit the accumulation of fatigue-related damage.
“Ninety percent of failure in metal components and engineering structures is through fatigue,” said Huajian Gao, a professor in Brown University’s School of Engineering and corresponding author of the new research. “This work represents a potential path to more fatigue-resistant metals, which would useful in nearly every engineering setting.”
Gao co-authored the study with Haofei Zhou, a postdoctoral researcher at Brown, along with Quingson Pan, Qiuhong Lu and Lei Lu from the Chinese Academy of Science.
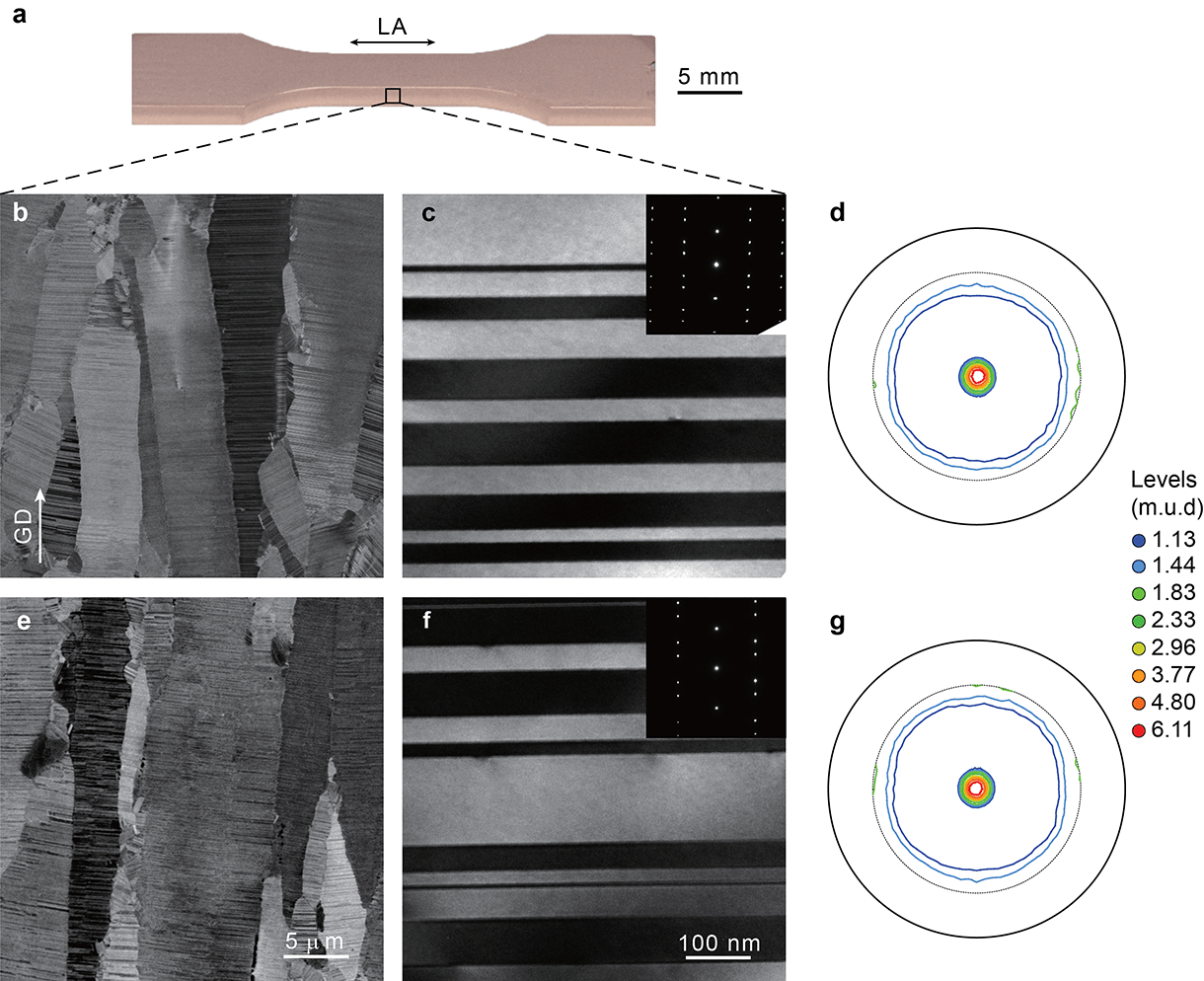
To study the fatigue effects of nanotwins, the researchers electroplated bulk samples of copper with closely spaced twin structures within the plates’ crystalline grains. Then they performed a series of experiments in which they stretched and compressed the plates repeatedly at different amplitudes of strain and measured the material’s associated stress response using a fatigue testing system. Beginning with a strain amplitude of .02 percent, the researchers progressively increased the amplitude every 1,500 cycles to .04, then .06, finally peaking at .09 before stepping back down through the strain amplitudes.
The tests showed that the stress response of the nanotwinned copper quickly stabilized at each strain amplitude. More importantly, Gao said, the study found that the stress response at each strain amplitude was the same during the second half of the experiment, when the metal was cycled through each strain amplitude a second time. That means the material did not harden or soften under the strain as most metals would be expected to do.
“Despite having already been through thousands of strain cycles, the material showed the same stress response,” Gao said. “That tells us that the reaction to cyclic strain is history-independent — the damage doesn’t accumulate the way it does in common materials.”
For comparison, the researchers performed similar experiments on non-nanotwinned samples, which showed significant hardening and softening (depending on the material) and displayed the type of cumulative fatigue effects that are common in most metals.
To understand the mechanism behind this fatigue resistance, the researchers performed supercomputer simulations of the metal’s atomic structure. At the atomic level, material deformation manifests itself through the motion of dislocations — line defects in the crystalline structure where atoms are pushed out of place. The simulations showed that the nanotwin structures organize strain-related dislocations into linear bands called correlated necklace dislocations (named for their beaded-necklace-like appearance in simulation). Within each crystal grain, the dislocations remain parallel to each other and don’t block each other’s motion, which is why the effects of the dislocations are reversible, Gao says.
“In a normal material, fatigue damage accumulates because dislocations get tangled up with each other and can’t be undone,” he said. “In the twinned metal, the correlated necklace dislocations are highly organized and stable. So when the strain is relaxed, the dislocations simply retreat and there’s no accumulated damage to the nanotwin structure.”
The metals aren’t entirely immune to fatigue, however. The fatigue resistance demonstrated in the study is within each crystalline grain. There’s still damage that accumulates at the boundaries between grains. But the within-grain resistance to fatigue “slows down the degradation process, so the structure has a much longer fatigue life,” Gao said.
Gao’s research group has worked extensively on nanotwinned metals, previously showing that nanotwin structures can improve a metal’s strength — the ability to resist deformation such as bending — and ductility, the ability to stretch without breaking. This new finding suggests yet another advantage to twinned metals. He and his colleagues hope this latest research will encourage manufacturers to find new ways of creating nanotwins in metals. The electroplating method used to fabricate the copper for this study isn’t practical for making large components. And while there are some forms of twinned metal available now (twinning-induced plasticity or “TWIP” steel is an example), scientists are still looking for cheap and efficient ways to make metals and alloys with twin structures.
“It’s still more of an art than a science, and we haven’t mastered it yet,” said Lu, one of the corresponding authors from Chinese Academy of Sciences. “We hope that if we point out the benefits you can get from twinning, it might stimulate fabrication experts to find new alloys that will twin easily.”
The work was supported by the U.S. National Science Foundation (DMR-1709318) and the National Natural Science Foundation of China. Computer simulation resources were provided by the U.S. NSF’s Extreme Science and Engineering Discovery Environment (XSEDE).