The University has a 5/3 day requirement for proposal submission. The completed proposal is due to OSP or BMRA five (5) business days before the sponsor deadline and the PI can take an additional three (3) days to finalize the scientific and/or programmatic components of the application. Guidance is presented below in four sections:
Timeline for scientific and/or programmatic components of the application
In order to allow Principal Investigators additional time for a final read‐through and last minute minor editing, corrections or updates to proposals, the five day requirement is amended as follows:
-
Complete proposals are due at OSP/BMRA five days before the sponsor’s deadline, however
-
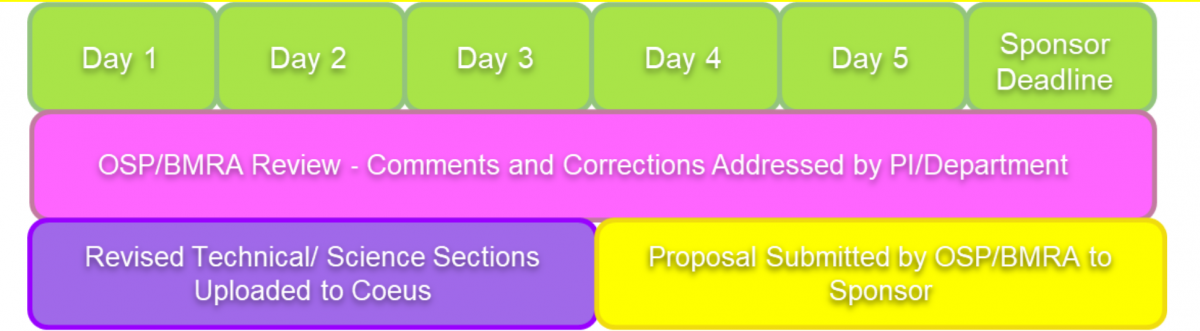
PIs will have until Day 3 of the 5 day period prior to the Sponsor’s due date to replace a draft version of technical/science documents with the corresponding final version in the Coeus proposal record. On the morning of Day 4 (8:30 am AY/8:00 am Summer) OSP/BMRA is authorized to submit the proposal without further review. PIs who take advantage of this relaxed internal deadline must accept the risk that a change to the technical section of the proposal may cause a submission failure. OSP/BMRA will not retrieve the proposal and correct resulting submission errors caused by PI changes to updated proposals.
-
The final version of a proposal will be submitted by OSP/ BMRA in advance of a Sponsor’s due date whenever possible to ensure it is successfully received by the sponsor.
In fairness to all Principal Investigators, on-time and complete proposals receive a thorough review and priority for submission.
Important notes to the above requirements (July 2023):
In rare and extenuating circumstances, when the University’s 5/3 day business day deadline cannot be met, a waiver may be requested. Approval is subject to departmental/center/institute/library and institutional review. Resources must be available locally to support proposal development. In such cases, an email waiver request from the Principal Investigator (PI) is sent to either the Senior Director, Office of Sponsored Projects (for all non-BioMed submissions and contract proposal submissions) or the Assistant Dean, Research Administration and Strategic Initiatives, Division of Biology & Medicine. A brief explanation as to the cause of the delay should be included. Our offices track these requests by PI name, department, and sponsor so we can monitor trends and offer support where needed.
There are times when a PI is informed of a potential funding opportunity within the 5 business days. An Agency’s Program Officer (PO) may reach out directly to the PI with late notice of available funds or a collaborator may send a late invitation to participate in the project. In these cases, a waiver from the 5/3 day rule is not required. Simply upload a copy of the PO or collaborators’ instructions to the proposal development record as this documentation will demonstrate a late request from the Sponsor. Then alert your departmental research administrator and the submitting office, OSP or BMRA.
Proposals which receive an expedited review will be given a less thorough review than those proposals which arrived in time to adhere to the University’s 5/3 Day timeline, as outlined above. The thoroughness of this expedited review will be dictated by several factors, including but not limited to University proposal volume, and the amount of time between routing and the sponsor’s deadline.
Submitting a Complete Proposal
The guidance requires that a complete proposal is due to OSP/BMRA five (5) days in advance of the sponsors’ due date. This allows routing in Coeus for Departmental approvals and the return of review comments from OSP/BMRA to the Department Managers and PIs. The submitted proposal must include all required elements specified in the sponsor’s application guidelines. Technical/Science sections submitted in draft format should be labeled in the document file title by section name and followed by “DRAFT” or “FINAL”.
If, on Day 1 a submitted proposal does not meet these criteria, the PI will be directed to the appropriate official to request a waiver of the 5 Day Rule.
During days 1‐3 PIs may devote extra time to “polishing” and a final read‐through of the technical section/ science. It is expected that the final science documents will differ only slightly (e.g., minor editing, figure captions, update to data tables, typo corrections) from the original version. OSP/BMRA will not review or provide feedback on ‘final’ technical sections.
Changes made to the scientific components must not affect any of the following elements:
-
Budget changes including direct and indirect costs; implied or stated cost‐sharing.
-
Involvement of non‐Brown employees; subcontractors; consultants and collaborators; foreign nationals or Federal employees; Export control concerns (ITARS/EARS);
-
Intellectual property (patents, copyrights, licenses); proprietary, privileged or potentially patentable information, conflicts of interest;
-
Involvement of human research participants, laboratory animal subjects and the environment, proposed clinical trials, or use of human embryonic stem cells;
-
Use of recombinant DNA, potentially infectious agents, hazardous waste, select agents, radiation producing or radioactive materials, biohazards;
-
Use of University facilities and equipment, additional space, alterations/construction, specialized or fabrication of equipment;
In the event an award is made and University obligations appear which were not present in the draft proposal, such obligations will be the sole responsibility of the PI and academic department.
Scientific Components of most common proposals submitted by Brown Faculty:
NIH: Project Summary, Research Plan – Specific Aims & Research Strategy, References Cited, Cover Letter for Resubmission applications
NSF: Project Summary, Project Description, Bibliography
Federal Prime Subawards: Scope of Work Statement
NASA: Proposal Summary (abstract), Scientific/Technical/Management Section, References and Citations
DOE: Project Description, Bibliography
DoD: Technical Approach and Justification, Project Schedule & Milestones
Practices for Monday due dates, holidays, and Pre-proposals
University policy states that proposals for external support of projects based at Brown are ordinarily due to the Office of Sponsored Projects (OSP) or BioMed Research Administration (BMRA) five (5) business days prior to the sponsor’s due date. However, the following exceptions are made to accommodate the proposal preparation process:
- For deadlines that fall on a MONDAY, complete proposals may be submitted until 9:00 am on the Monday prior to the deadline date. This gives the PI additional weekend to work through the technical portions of the application.
-
For University Holidays that fall on Monday, proposals are due to OSP/BMRA on TUESDAY by noontime after the Holiday. The Holidays include
- Labor Day First Monday in September
- Fall Weekend (observed) Second Monday in October
- Martin Luther King, Jr. Day (obs.) Third Monday in January
- Memorial Day Last Monday in May
Further Guidance on the 5/3 Day Rule by Proposal Type
White paper or Concept Paper - preliminary ideas or data presented to determine interest of sponsor in funding a project or program
Short narrative (1-5 pages)
No OSP/BMRA review, PI submits directly to sponsor
5/3 day proposal submission rule does not apply
Letter of Intent to apply (a/k/a "LOI") - informs sponsor of planned full proposal
Short narrative (1-5 pages)
Total cost estimate only, no detailed budget
OSP/BMRA approval is optional (per Sponsor instructions)
No COEUS record, PI or designee submits to sponsor
5/3 day proposal submission rule does not apply
Pre‐Proposal (simple) - federal and non-federal sponsors
Narrative with minimal other components (e.g., Biosketch, COA List)
Total cost estimate, no detailed budget
OSP/BMRA approval is optional (per Sponsor instructions)
No COEUS record
PI or designee submits directly to sponsor
5/3 day proposal submission rule does not apply
Pre‐Proposal (complex) - National Science Foundation (NSF), other federal and non-federal sponsors
Narrative with minimal other components (e.g., Biosketch, COA List)
Detailed budget may be required
OSP/BMRA approval may be required
COEUS record required
Research.gov/Fastlane and federal submission by OSP/BMRA
Non-federal sponsor submission may be by PI or Designee, OSP/BMRA
Due to OSP/BMRA 3 days before Sponsor's due date
Full Proposal
Narrative with statement of work; detailed budget and justification; current/pending support; biosketch/cv; facilities and environmental; bibliography/references, ect.
Detailed line item or modular budget is required
OSP/BMRA signature required
Coeus record required
Submission by OSP/BMRA
Due to OSP/BMRA 5 days before Sponsor's due date
Notes
- If award terms or conditions are included in sponsor's instructions, approval, review and submission by OSP / BMRA is mandatory.
- If sponsor restricts the number of applicants from Brown University, advance clearance from OVPR’s Office of Research Strategy and Development is required.
- If there is any potentially patentable information, include markings that designate the information as confidential/ proprietary. Principal Investigators who are concerned about protecting intellectual property as either patent or copyright, are encouraged to contact Brown Technology Innovations.
Please also see the Proposal Submission Guidance Chart for further guidance on which proposals require a Coeus record
Sept 1, 2009
Dear Colleagues,
We write to request your cooperation and assistance in Brown University's commitment to meeting agency deadlines while submitting proposals of the highest quality, and to maintain fairness and consistency among faculty colleagues in this process.
For some time we have had a policy requesting that PIs submit proposals to OSP/BMRA five days before the agency deadline, in order to allow for adequate review, correction of errors or omissions, compliance with agency requirements, and submission through a variety of electronic mechanisms. The five day rule has not been widely observed and our experience over the last year has included several large proposals which nearly missed the agency deadline and numerous serious errors in proposals that caused them to be returned. These events were directly attributable to the lateness of their submission to OSP or BMRA.
Furthermore, an increasingly unfair burden is placed upon department administrators who are tasked with submitting late and incomplete proposals to OSP/BMRA, and upon principal investigators who do submit their proposals on time only to see them put aside while late proposals are processed as emergencies. These events have convinced us that the five day rule must now be made mandatory.
Therefore, effective October 1, we are instructing OSP and BMRA NOT TO ACCEPT FOR REVIEW ANY PROPOSAL THAT IS LATER THAN 5 BUSINESS DAYS PRIOR TO THE AGENCY DEADLINE FOR SUBMISSION. For example, proposals intended to be submitted for the upcoming October 5 agency deadlines, that are received in OSP or BMRA after the close of business on September 25, will be returned to the PI without further action. It is incumbent upon our staffs to abide by this policy, and exceptions are not anticipated.
In the case of serious extenuating circumstances, a request for exception to this policy must be made at least five days before the deadline directly to Clyde Briant, who will consult with Ed Wing as appropriate.
We appreciate your understanding of the seriousness of this issue, and for supporting our efforts to provide comprehensive, substantive, and productive assistance to all faculty submitting proposals for external funding.
Sincerely,
Dr. Clyde Briant
Vice President for Research
Dr. Edward Wing
Dean of Medicine & Biological Sciences
